Optical activity and specific rotation
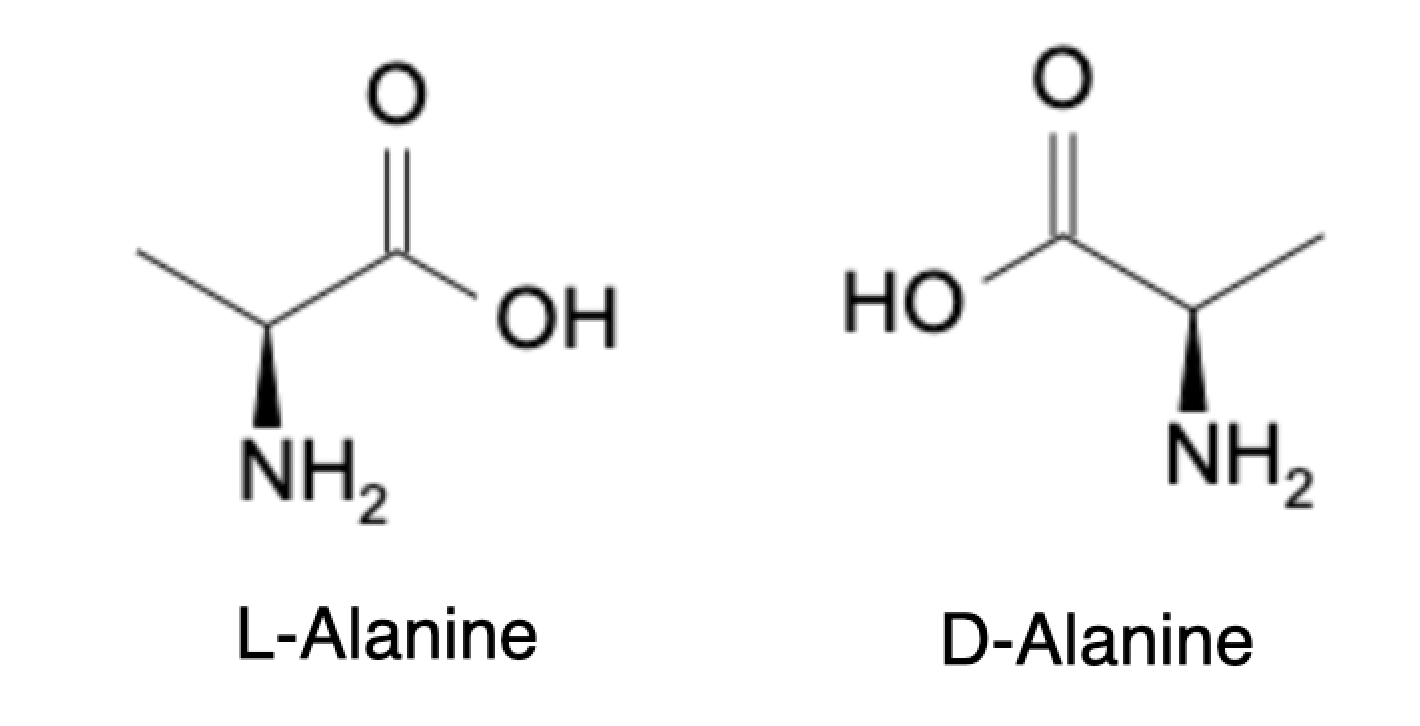
In organic chemistry, chirality refers to the property of a molecule that cannot be superimposed on its mirror image. Molecules that possess chirality are called chiral molecules, and they exist in two mirror image forms known as enantiomers. The two enantiomers have the same physical and chemical properties, except that they interact differently with polarized light, which is referred to as optical activity.
Optical activity
Optical activity occurs when polarized light passes through a chiral molecule and is rotated by an angle, either clockwise or counterclockwise. The direction of rotation depends on the configuration of the molecule, specifically whether it is a dextrorotatory (rotates polarized light to the right) or levorotatory (rotates polarized light to the left) enantiomer. The degree of rotation is determined by the concentration of the enantiomer in solution, the length of the sample tube, and the wavelength of the light used.
The magnitude of the optical rotation is quantified by the specific rotation, which is a property of the enantiomer and is denoted by the symbol (α). The specific rotation is defined as the observed rotation (in degrees) divided by the concentration (in g/mL) and the length of the sample tube (in dm). It is a constant value for a given enantiomer at a specific temperature and wavelength of light.
Racemic Mixtures
Some chiral molecules are optically inactive, meaning that they do not rotate polarized light. This is because they exist as a racemic mixture, which is a mixture of equal amounts of both enantiomers. A racemic mixture has no net optical activity because the rotations of the two enantiomers cancel each other out.
It is important to note that not all molecules with stereocenters are chiral. Molecules with an internal plane of symmetry, also known as meso compounds, have an identical mirror image and are optically inactive. This is because they have both enantiomers present within the same molecule and their rotations cancel out.
Summary
Optical activity is a property of chiral molecules that can be used to distinguish between their enantiomers. The specific rotation is a measure of the degree of optical activity and is a constant value for a given enantiomer at a specific temperature and wavelength of light. Meso compounds, which have an internal plane of symmetry, are optically inactive and do not exhibit optical activity.
Test Your Knowledge:
What is the difference between a chiral and an achiral molecule?
How is the specific rotation of an enantiomer determined?