Corey–House (Gilman) Coupling — R₂CuLi with Alkyl Halides
Lithium diorganocuprates (Gilman reagents, R₂CuLi) couple with suitable alkyl halides (R′–X) to forge new C–C σ bonds (output: R–R′). In undergraduate synthesis, this is the go‑to way to join a primary, allylic, or benzylic sp³ electrophile with an aliphatic/aryl fragment carried by copper. Only one “R” transfers from R₂CuLi; the byproduct is RCu (plus LiX). The most common teaching picture is: Cu(I) associates with R′–X → formal oxidative addition to Cu(III) → reductive elimination to deliver R–R′ with inversion at R′ (SN2‑like). Typical conditions: make R₂CuLi from RLi + CuI in dry Et₂O/THF (−78 → 0 °C), then add R′–X (X = I, Br; Cl is slower). Alkyl fluorides and tertiary halides fail.
Need a metal-carbon partner? Compare with acid chloride + Gilman (ketone) or Alkyl halide → Grignard formation when you need more basic nucleophiles.
Quick Summary
- Reagents/conditions: Prepare R₂CuLi (2 R–Li + CuI) in dry Et₂O/THF at −78 °C → 0 °C; add R′–X (I ≥ Br ≫ Cl; tosylates work). Quench with NH₄Cl or dilute acid to remove copper salts.
- Electrophile scope: Primary (best), allylic, benzylic halides/sulfonates. Secondary = sluggish with E2; tertiary = elimination/no coupling. Alkyl fluorides are effectively inert.
- Outcome: R–R′ (only one “R” from the cuprate transfers). Byproducts: RCu and LiX.
- Stereochemistry: Inversion at chiral primary R′–X is observed (SN2-like). Allylic partners frequently give SN2′ products (allylic transposition).
- Mechanistic teaching model: Cu(I) associates → formal Cu(III) oxidative addition → reductive elimination → Cu(I) + R–R′.
Mechanism — Association → Reductive Elimination (Mechanism Solver Frames)
Teaching note: The Mechanism Solver shows two frames. The classical “Cu(I) → Cu(III) → Cu(I)” picture is implied—association builds the ate complex and oxidative addition, then reductive elimination couples R–R′ and regenerates Cu(I).
Step 1 — Association / Ate Complex Formation
Step 2 — Reductive Elimination (C–C Bond Formation)
Mechanistic Checklist
- Electrophile class: Primary (including benzylic/allylic) works best; secondary is low‐yield; tertiary fails.
- Leaving group: I ≥ Br > Cl ≫ F. Tosylates/mesylates behave like bromides.
- Only one transfer: Each R₂CuLi delivers a single “R”; the partner R remains as RCu byproduct.
- Stereochemistry: Expect inversion at a chiral primary center (SN2-like). Allylic halides often yield SN2′ products.
- Moisture/air sensitive: Make and use R₂CuLi in dry, inert ether at low temperature.
- Compare to Grignards: Cuprates are softer/less basic → reduced elimination and improved functional-group tolerance.
Worked Examples
Each card uses the exact SMILES fed to the Mechanism Solver (reactant SVG) plus the exported product frame from the RDKit backend. The Gilman button is the same art that appears in the toolbox, making it easy to cross-reference.
MeI + n-Bu₂CuLi → Pentane
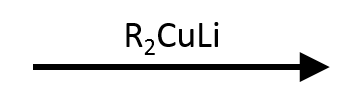
Methyl iodide undergoes a simple SN2-like coupling: one n-butyl fragment transfers while the second stays on copper (RCu byproduct).
Allyl bromide + (Ph)₂CuLi → Allylbenzene (SN2′)
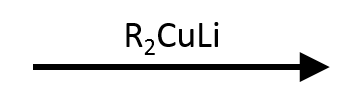
Allylic substrates frequently deliver the transposed SN2′ product; the Mechanism Solver highlights the distal carbon coupling.
(S)-2-bromobutane + (Cy)₂CuLi → (R)-cyclohexylbutane
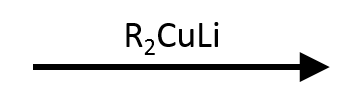
Secondary substrates can couple but require cold conditions; note the inversion at the stereocenter and the warning about competing elimination.
tert-Butyl bromide → Isobutene (Failure)
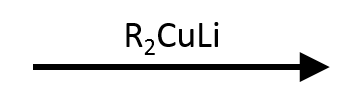
Tertiary halides do not participate; β-elimination dominates, which is why the mechanism builder issues an upfront warning.
Scope & Limitations
- Works well: Primary alkyl iodides/bromides/chlorides; benzyl and allyl halides; tosylates/mesylates.
- Moderate: Secondary halides (low yields, elimination). Chlorides are slower but manageable.
- Fails under this manifold: Tertiary halides, alkyl fluorides, and (in this course) vinyl/aryl halides (teach Pd cross-couplings instead).
- Cuprate fragment R: Alkyl, aryl, and α-heteroatom stabilized chains are compatible. Consider R–Li availability.
Practical Tips & Pitfalls
- Make cuprate in situ: 2 R–Li + CuI at −78 °C, then add R′–X slowly.
- Exclude air/moisture: Organocuprates are air- and water-sensitive—use dry glassware, inert gas, cold temperatures.
- Order of addition: Often add the electrophile to the cuprate (not vice versa) to minimize R′–X decomposition.
- Plan stoichiometry: One equivalent of R₂CuLi transfers one R fragment.
- Use the right leaving group: Iodides/bromides or sulfonates; avoid fluorides unless a different coupling (e.g., Pd) is specified.
- Highlight byproducts: Mention RCu and LiX in mechanism frames or exam answers.
Exam-Style Summary
Corey–House: 2 R–Li + CuI → R₂CuLi; R₂CuLi + R′–X → R–R′ (best with primary/allylic/benzylic halides; X = I/Br/Cl). Only one R transfers. Mechanistic shorthand: Cu(I) → Cu(III) oxidative addition, then reductive elimination (net SN2/inversion at R′). Secondary = low yield (E2); tertiary/alkyl fluorides = no coupling.
Interactive Toolbox
- Mechanism Solver — Visualize the OA → RE frames (primary vs allylic vs blocked electrophiles).
- Reaction Solver — Practice identifying when R₂CuLi succeeds (primary/allylic/benzylic) vs fails.
- IUPAC Namer — Confirm product names like allylbenzene or cyclohexylpropane.
Try the Mechanism Solver with “R₂CuLi / Alkyl halide” and compare primary vs secondary vs allylic outcomes.