Esters + NaOR: Transesterification vs Claisen
NaOR reagents toggle between two archetypal ester pathways:
- Base-catalyzed transesterification (OR swap): catalytic NaOR in the matching ROH swaps –OR′ for –OR without forming new C–C bonds. Equilibrium-driven; push with excess ROH or removal of R′OH.
- Claisen condensation (β-keto ester formation): stoichiometric NaOR (OR matches the ester’s leaving group), requires an α-H, and ends as an enolate until H₃O⁺ workup delivers the β-dicarbonyl product.
Exam pivots: Match OR for Claisen to avoid scrambling, remember the Claisen enolate consumes base until quench, and treat transesterification as an equilibrium process controlled by Le Châtelier (excess ROH or distilling R′OH).
In the app, the Reaction Solver offers separate buttons for the catalytic OR-swap path and for the Claisen path. The Claisen option automatically clones the OR group directly from your drawing into the NaOR overlay, so you can stress-test nonstandard alkoxides or conceptual cases without extra setup.
Seafoam teal Color 1 (RGB 93,190,197; hex #5DBEC5) now highlights the incoming OR fragment in every mechanism panel and worked example so you can follow that group from reagent badge to product instantly. When a second fragment needs its own visual trace, Golden Peach Color 2 (RGB 244,199,127; hex #F4C77F) will tag that add-on in parallel, with both tones called out in captions and alt text.
Quick Summary
| Pathway | Conditions | Outcome |
|---|---|---|
| Transesterification | Cat. NaOMe/MeOH or NaOEt/EtOH, excess ROH, rt–reflux. | OR′ exchange only (R′COOR″ → R′COOR). Equilibrium; no net C–C change. |
| Claisen condensation | ≥1 eq NaOR (OR matches ester OR′), an α-H-bearing ester, ROH solvent, then H₃O⁺. | β-keto ester (or cyclic Dieckmann) plus expulsion of OR′. Requires α-H and matching OR. |
Mechanism — Path A: Transesterification
Transesterification is catalytic in base and reversible.
- RO⁻ attack: NaOR (dissolved in its matching ROH) adds to the ester carbonyl to form a tetrahedral alkoxide.
- Collapse: The carbonyl reforms and expels the original OR″ as R″O⁻.
- Catalyst regeneration: Proton transfers between ROH and R″O⁻ regenerate RO⁻. Drive the equilibrium with excess ROH or by removing R″OH.
Keep NaOR loadings catalytic (≈5–10 mol%) and maintain anhydrous conditions to avoid saponification.
Mechanism — Path B: Claisen Condensation
Claisen condensation requires stoichiometric NaOR, at least one α-hydrogen, and ends as an enolate until workup.
- Enolate formation: NaOR—now drawn with the exact OR chain from your substrate—deprotonates the α-carbon.
- C–C bond formation: The enolate attacks a second ester, creating a tetrahedral adduct.
- Collapse & OR departure: The adduct shows both esters: O⁻ collapses back to C=O as the newly attached OR group leaves.
- Acidic workup: H₃O⁺ protonates the β-dicarbonyl enolate to give the neutral β-keto ester (or cyclic Dieckmann product) with the full electrophile carbon chain.
Dieckmann condensation follows the same logic intramolecularly; it remains outside this button’s scope.
Step-by-Step Comparison
| Transesterification | Claisen Condensation |
|---|---|
|
Step 1 — RO⁻ attack. |
Step 1 — Enolate formation. |
|
Step 2 — Collapse. |
Step 2 — C–C bond formation. |
|
Step 3 — Proton transfers regenerate RO⁻. |
Step 3 — Collapse & OR departure. |
|
Transesterification stops here (neutral ester product). |
Step 4 — H₃O⁺ workup. |
Mechanistic Checklist (Exam Focus)
- Transesterification: Show RO⁻ addition → tetrahedral → collapse → catalyst regeneration; emphasize equilibrium control (excess ROH or distill R″OH). Avoid confusing NaOR with NaOH (saponification).
- Claisen: α-H required; the app now clones your exact OR group into the NaOR overlay, so you can explore theoretical OR variants, but the classic best practice is matching OR′ (the UI emits a warning when you mismatch). Depict enolate generation → addition → collapse/OR departure → H₃O⁺ quench.
- Dieckmann: Same logic intramolecular (5–6 membered rings most favorable). Currently outside the button’s scope but mechanistically analogous.
- Mixed Claisen: Use one non-enolizable ester (e.g., aryl formate) so only the α-H-bearing partner forms the enolate; the solver lets you toggle
hasAlphaHto test feasibility.
Worked Examples
| Scenario | Inputs | Key Output / Highlight |
|---|---|---|
| Example 1 — Transesterification | Methyl isobutyrate + NaOCy (C₆H₁₁ONa) in cyclohexanol, catalytic base, excess ROH. | Cyclohexyl isobutyrate; seafoam-teal Color 1 shows the incoming cyclohexyloxy group installed by NaOCy. |
| Example 2 — Claisen condensation | Ethyl acetate + NaOEt (1.1 eq) in EtOH; β-dicarbonyl enolate quenched with H₃O⁺. | Ethyl acetoacetate; seafoam-teal Color 1 traces the β-dicarbonyl arm formed during C–C bond construction. |
Reactant
Methyl isobutyrate
Reagent
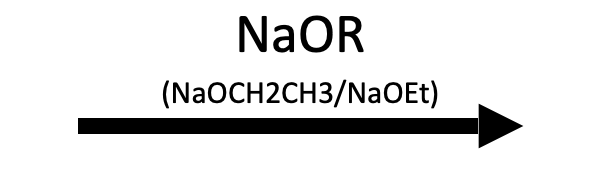
NaOCy (seafoam teal Color 1 = incoming OR)
Product
Cyclohexyl isobutyrate (seafoam teal Color 1 CyO−)
Reactant
Ethyl acetate
Reagents

NaOEt (seafoam teal Color 1 EtO⁻) and EtOH solvent
Product
Ethyl acetoacetate (seafoam-teal Color 1 β-dicarbonyl)
Scope, Selectivity & Edge Cases
- Transesterification: Equilibrium-limited; best when R′OH is volatile/removed. Avoid strong base loading or α-H activation if Claisen is undesired.
- Claisen: Needs α-H; literature strongly prefers matching the base OR to the leaving group (NaOEt with ethyl esters). The solver lets you experiment with mismatches, but expect warnings and lower yields in real life. Bulky esters react slower; mixed Claisen works when one partner lacks α-H.
- Dieckmann: 5–6 member rings favored; 4- or 7+ membered rings are disfavored due to ring strain/entropic cost.
- Functional group tolerance: Both manifolds are strongly basic. Protect acid-sensitive moieties (e.g., carbonyls prone to enolate formation) accordingly.
Practical Tips & Pitfalls
- Transesterification: Use ~5–10 mol% NaOR in the matching ROH. Apply Dean–Stark or gentle distillation to remove R″OH. Monitor to avoid unintended Claisen if the ester has α-H and the base accumulates.
- Claisen: Use ≥1 eq NaOR, matching OR′. Cool during base addition; only add the second ester once the enolate forms. Quench with cold H₃O⁺ to avoid self-transesterification of the β-keto ester.
- Avoiding saponification: Water or NaOH shifts to irreversible hydrolysis (different mechanism). Keep conditions anhydrous unless the endpoint is hydrolysis.
Exam-Style Summary
| Condition | Expectation |
|---|---|
| Cat. NaOR in ROH + excess ROH | Transesterification (OR swap). |
| Stoichiometric NaOR (matched) + α-H ester + H₃O⁺ | Claisen condensation → β-keto ester (or cyclic Dieckmann). |
| No α-H or mismatched base | Claisen fails; default to OR exchange or no reaction. |
FAQ
Does Claisen absolutely require the base OR to match the ester’s OR′?
That’s the gold-standard condition in textbooks because it keeps the leaving group and the base conjugate acid identical, minimizing scrambling. The solver lets you test mismatched cases for curiosity, but expect warnings (and messy mixtures in practice).
Can I explore hypothetical alkoxides (allylic, benzylic, etc.)?
Yes. Whatever OR group you draw on the ester will be cloned into the NaOR overlay for the Claisen pathway, so you can visualize truly bespoke alkoxides for conceptual checks.
What if my ester lacks an α-hydrogen?
Claisen formation stalls because the first step (enolate formation) fails. Use the Reaction Solver’s hasAlphaH toggle to see the error message, or pair the non-enolizable ester with an α-H partner in a mixed Claisen setup.
Is Dieckmann condensation included in this button?
Not yet. The current build handles intermolecular Claisen only. The intramolecular Dieckmann pathway follows the same logic but needs additional ring-size heuristics; it’s flagged here as a conceptual cousin but isn’t automated.
Interactive Toolbox
- Mechanism Solver animates either pathway; the Claisen button clones your OR group into the NaOR overlay so you can compare branches with the same substrate.
- Reaction Solver flips between NaOMe/MeOH and NaOEt/EtOH, toggles the α-H flag, and previews whether conditions lead to OR swap or β-keto ester.
- IUPAC Namer captions outputs (methyl acetate, ethyl acetoacetate, etc.) without exposing SMILES strings.