Oxidation and Reduction in Organic Chemistry
Every reagent, when to use it, and how to pick the right one on an exam
Oxidation and Reduction in Organic Chemistry
Oxidation and reduction in organic chemistry works differently than what you learned in gen chem. Forget oxidation numbers for individual atoms, forget half-reactions, forget electrochemical cells. In organic chemistry, you need to look at carbon and count its bonds.
Once you see it the right way the logic is surprisingly simple. This guide covers how to recognize oxidation vs. reduction at a glance, every major reagent (and which ones are selective), and the decision logic for picking the right one on an exam.
How Oxidation State Works in Organic Chemistry
In gen chem, you tracked electrons. In organic, you track bonds.
Oxidation = carbon forms more bonds to oxygen (or other electronegative atoms), OR carbon loses bonds to hydrogen.
Reduction = carbon forms more bonds to hydrogen, OR carbon loses bonds to oxygen.
Every oxidation and reduction reaction in your entire organic chemistry course follows this pattern.
The oxidation state ladder for a one-carbon compound looks like this:
CH₄ → CH₃OH → CH₂O → HCO₂H → CO₂ (methane → methanol → formaldehyde → formic acid → carbon dioxide)
Each step to the right is an oxidation (losing C-H bonds, gaining C-O bonds). Each step to the left is a reduction. You can go up one rung at a time, or skip rungs, depending on your reagent.
For the functional groups you actually see on exams, the ladder that matters is:
Alkane → Alcohol → Aldehyde/Ketone → Carboxylic Acid
Or for the alcohol-focused version that gets tested the most:
| Starting Material | Oxidation Product | Reagent Type Needed |
|---|---|---|
| 1° Alcohol | Aldehyde (selective) OR Carboxylic acid (full) | Mild oxidant OR Strong oxidant |
| 2° Alcohol | Ketone | Any oxidant (ketone can't oxidize further under normal conditions) |
| 3° Alcohol | No reaction | — (no hydrogen on the carbinol carbon to remove) |
| Aldehyde | Carboxylic acid | Most oxidants |
| Ketone | No reaction (under normal conditions) | — |
The 3° alcohol entry is the one professors test. If you see a tertiary alcohol treated with any oxidizing agent, the answer is no reaction. There's no hydrogen on the carbon bearing the -OH, so the mechanism can't proceed.
Quick Check: A 3° alcohol is treated with PCC. What happens?
Reveal answer
No reaction. PCC oxidizes alcohols by removing the hydrogen on the carbinol carbon (the carbon bearing the -OH). A tertiary alcohol has no hydrogen there. The carbinol carbon is bonded to three other carbons and the -OH. It doesn't matter which oxidizing agent you use. If the alcohol is tertiary, the answer is always no reaction.
Quick recognition trick: Count the number of C-H bonds on the carbon that changes. If that number goes down, it's an oxidation. If it goes up, it's a reduction. If C-H count stays the same, it's neither.
3° alcohols don't oxidize. The carbinol carbon has no hydrogen to remove, and every standard oxidation mechanism needs that C-H. Any oxidizing agent + 3° alcohol = no reaction.
The Reagents: Oxidizing Agents OXIDIZE
Strong Oxidizing Agents (Go All the Way)
These oxidize 1° alcohols directly to carboxylic acids, skipping the aldehyde. They'll also oxidize 2° alcohols to ketones. Use them when you want the full oxidation, not a partial one.
Jones Oxidation. CrO₃ / H₂SO₄ / H₂O (or Na₂Cr₂O₇ / H₂SO₄) 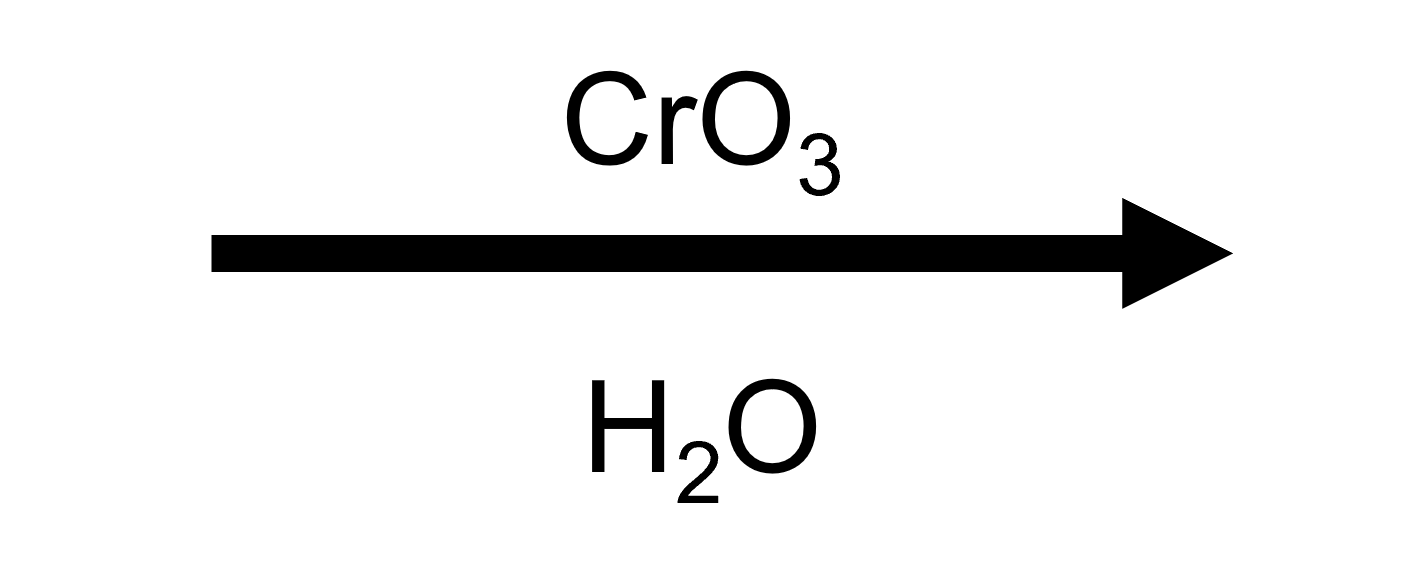
The classic. Chromium(VI) in aqueous acid. Oxidizes 1° alcohols all the way to carboxylic acids. Oxidizes 2° alcohols to ketones. No reaction with 3° alcohols.
Jones goes all the way to the carboxylic acid because of the aqueous conditions. The aldehyde intermediate, once formed, doesn't survive. Water attacks the aldehyde carbonyl, forming a geminal diol (hydrate). That hydrate now has a C-H and an -OH on the same carbon. Exactly the setup of a secondary alcohol, which chromium(VI) oxidizes again to give the carboxylic acid. The aldehyde never accumulates because it keeps getting hydrated and re-oxidized.
PCC avoids this by running in anhydrous CH₂Cl₂. No water, no hydration, no second oxidation. The selectivity isn't about the strength of the oxidant; it's about whether water is present to enable the second step.
The downside: chromium is toxic, and the aqueous acidic conditions aren't compatible with acid-sensitive functional groups elsewhere in the molecule.
KMnO₄ (potassium permanganate) 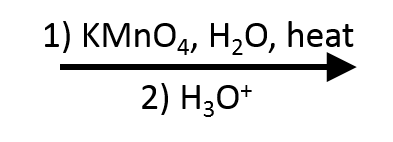
Another strong oxidant. Hot, concentrated, or acidic KMnO₄ oxidizes 1° alcohols to carboxylic acids. Also oxidizes alkenes (see below). Cold, dilute, basic KMnO₄ is milder and does syn-dihydroxylation of alkenes instead. Context matters with this reagent.
What to do when you don't recognize the reagent: Exams sometimes include reagent variants not on the standard list (IBX, PDC, Pb(OAc)₄). If you see an unfamiliar oxidizing agent, look for these signals:
- Chromium(VI) in any form (CrO₃, CrO₄²⁻, Cr₂O₇²⁻, PDC, PCC): oxidizes alcohols. Aqueous = goes to carboxylic acid. Anhydrous = stops at aldehyde.
- Permanganate (MnO₄⁻): strong oxidant. Hot/acidic = cleaves alkenes, oxidizes alcohols to acids. Cold/dilute/basic = syn dihydroxylation.
- Peracids (RCO₃H, anything with "-peroxy-" or "-peracid"): epoxidizes alkenes. Does not oxidize alcohols.
- OsO₄ or RuO₄: dihydroxylation of alkenes.
- Any metal hydride (H⁻ donor): that's a reducing agent, not an oxidant. Boron hydrides are mild (NaBH₄), aluminum hydrides are strong (LiAlH₄, DIBAL-H).
- H₂ + metal catalyst: hydrogenation. Reduces pi bonds.
Selective (Mild) Oxidizing Agents (Stop at the Aldehyde)
These are the reagents that let you oxidize a 1° alcohol to an aldehyde WITHOUT going further to the carboxylic acid. This is the selectivity question professors test constantly: "How would you convert 1-butanol to butanal?"
If you answer "Jones oxidation" or "KMnO₄," you just blew past the aldehyde to the carboxylic acid. You need a mild, selective oxidant.
PCC. Pyridinium Chlorochromate (C₅H₅NH⁺ CrO₃Cl⁻) 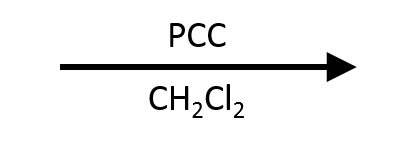
The most common selective oxidation reagent. Operates in anhydrous CH₂Cl₂ (no water), so the aldehyde product cannot hydrate and survives without a second oxidation.
PCC oxidizes:
- 1° alcohol → aldehyde (STOPS HERE)
- 2° alcohol → ketone
- 3° alcohol → no reaction
This is probably the single most-tested selective oxidation reagent in organic chemistry.
DMP. Dess-Martin Periodinane 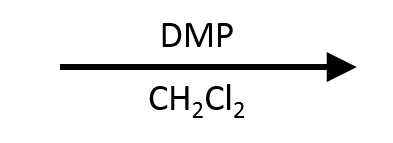
A modern alternative to PCC. Same selectivity (stops at aldehyde for 1° alcohols), but milder conditions, easier workup, and no toxic chromium waste. Does the same job as PCC through an iodine-based mechanism instead.
DMP oxidizes:
- 1° alcohol → aldehyde (STOPS HERE)
- 2° alcohol → ketone
Swern Oxidation. (COCl)₂ / DMSO / Et₃N
Another selective option. Uses oxalyl chloride and DMSO at low temperature (-78°C), followed by triethylamine. Stops at the aldehyde for 1° alcohols. If an exam shows DMSO and (COCl)₂ together, or calls out -78°C, it's a Swern.
How to remember: PCC, DMP, and Swern all do the same thing. Stop at the aldehyde. They differ in mechanism and practical considerations, but for product prediction on exams, they're interchangeable. PCC is the one you'll see most often.
Whether your professor expects you to know DMP and Swern by name or just PCC depends entirely on the course. Check your syllabus. If they've only shown you PCC in lecture, that's probably the only selective oxidant they'll test.
The Selectivity Decision in One Line
Want a carboxylic acid from a 1° alcohol? → Jones (CrO₃/H₂SO₄/H₂O) or KMnO₄. Want an aldehyde from a 1° alcohol? → PCC, DMP, or Swern. Oxidizing a 2° alcohol to a ketone? → Any of them work. Ketones can't be oxidized further under these conditions, so the reagent choice rarely matters. Pick whichever was shown in lecture.
Jones vs PCC comes down to water. Jones is aqueous → the aldehyde hydrates and re-oxidizes → carboxylic acid. PCC/DMP/Swern are anhydrous → no hydrate → aldehyde survives. Same selectivity logic for every 1° alcohol oxidation problem.
One more selectivity question shows up on exams: what if the molecule has both an alcohol and an alkene? KMnO₄ and OsO₄ oxidize both, and Jones also hits both under forcing conditions.
PCC is the answer here: it oxidizes alcohols but doesn't touch C=C double bonds. If your substrate has an alcohol you want to oxidize and an alkene you want to preserve, PCC in CH₂Cl₂ is the only standard reagent that gives you that selectivity.
What about two different alcohols in the same molecule? If a substrate has both a 1° and a 2° alcohol, PCC, Jones, and KMnO₄ all hit both without distinguishing. You'd need a protecting-group strategy: protect one, oxidize the other, deprotect.
Alcohol + ester is easier: PCC and Jones oxidize the alcohol without touching the ester. The oxidation selectivity story on exams is almost always alcohol-vs-alkene or strong-vs-mild, not discriminating between two alcohols.
Quick Check: You need to convert 1-hexanol to hexanal. You reach for Jones reagent. What's wrong with that choice?
Reveal answer
Jones reagent uses aqueous conditions, which let the aldehyde hydrate and get oxidized a second time. You'd end up with hexanoic acid, not hexanal. For the aldehyde, you need a selective oxidant. PCC in CH₂Cl₂, DMP, or Swern. The anhydrous conditions protect the aldehyde from further oxidation.
The Reagents: Reducing Agents REDUCE
Reduction is the reverse direction: adding hydrogen, removing oxygen, converting C=O back to C-OH or C-H.
NaBH₄: Sodium Borohydride 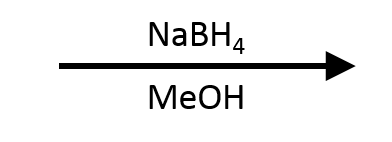
The mild reducing agent. Delivers H⁻ (hydride) to electrophilic carbons.
NaBH₄ reduces:
- Aldehydes → 1° alcohols
- Ketones → 2° alcohols
- Does NOT reduce carboxylic acids, esters, or amides
That last line is critical. NaBH₄ is selective. It only reduces aldehydes and ketones. If your substrate has both a ketone and an ester, NaBH₄ reduces the ketone and leaves the ester untouched. This selectivity gets tested.
Solvent: typically MeOH or EtOH (protic solvents are fine because NaBH₄ is mild enough to tolerate them).
One note on α,β-unsaturated carbonyls (enones): NaBH₄ typically gives 1,2-reduction at the carbonyl even when a C=C is conjugated with the C=O. For clean 1,4- (conjugate) reduction of an enone, the standard reagents are L-selectride, Stryker's reagent [(Ph₃P)CuH]₆, or NaBH₄ with CuCl. Don't confuse this with Gilman cuprates (R₂CuLi), which do conjugate addition of a carbon nucleophile, not hydride delivery. If your professor covers Luche conditions, CeCl₃/NaBH₄ ensures clean 1,2-selectivity for substrates where it might be ambiguous.
LiAlH₄: Lithium Aluminum Hydride 
LiAlH₄ reacts violently with water. It's a strong enough base and nucleophile to deprotonate protic solvents on contact, which means you can never run it in water, methanol, or ethanol. The solvent is always anhydrous ether (Et₂O or THF), and the aqueous acid workup (H₃O⁺) comes after the reaction is done. That's what the two-step notation "(1) LiAlH₄/Et₂O, (2) H₃O⁺" refers to.
LiAlH₄ also reduces almost every carbonyl and strained C-O system it meets: aldehydes, ketones, carboxylic acids, esters, amides, epoxides. Esters give two alcohol products (one from the acyl side, one from the alkoxy side). Amides give amines. It's powerful but indiscriminate, so if selectivity matters you need a milder reagent.
DIBAL-H: Diisobutylaluminum Hydride 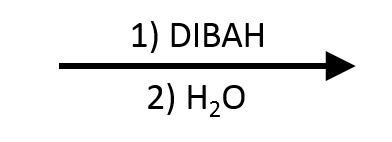
DIBAL-H exists to solve one specific problem that neither NaBH₄ nor LiAlH₄ can handle: reducing an ester to an aldehyde and stopping there.
This transformation is hard because NaBH₄ can't even get started (esters are below its reactivity threshold), and LiAlH₄ is powerful enough to reduce the ester but doesn't stop at the aldehyde. The aldehyde is actually more reactive than the ester, so LiAlH₄ blows right through it to the alcohol. The reagent you need is strong enough to start the reduction but controllable enough to stop partway.
That's DIBAL-H at -78°C with one equivalent. At that temperature, DIBAL-H reduces the ester to a tetrahedral aluminum alkoxide intermediate. The cold temperature stabilizes that intermediate. It doesn't collapse further until aqueous workup, at which point it gives the aldehyde. Warm it up or add excess DIBAL-H, and it keeps going to the alcohol, just like LiAlH₄.
DIBAL-H works poorly on free carboxylic acids: the acidic -COOH proton reacts with DIBAL-H first in an acid-base step, wasting an equivalent of reagent before any reduction starts, and the typical outcome is either incomplete conversion or overshoot to the alcohol. Treat esters as the standard substrate for this transformation. If you need an aldehyde from a carboxylic acid, a cleaner route is converting it to the acid chloride (SOCl₂) first and then running a Rosenmund-style reduction.
Temperature is the giveaway for DIBAL-H. A reduction problem that calls out "-78°C" on an ester is almost always testing this reagent.
DIBAL-H at -78°C is the only standard way to stop ester reduction at the aldehyde. NaBH₄ is too weak to start, LiAlH₄ overshoots to the alcohol. Cold DIBAL-H traps the tetrahedral intermediate until workup.
Catalytic Hydrogenation: H₂ / Pd, Pt, or Ni 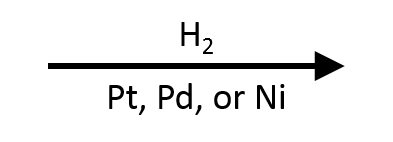
A completely different approach to reduction. Instead of hydride delivery, you're adding H₂ across a pi bond using a metal catalyst.
H₂/Pd (or Pt, or Ni) reduces:
- Alkenes → alkanes (syn addition of H₂)
- Alkynes → alkenes (with Lindlar's catalyst, stops at cis-alkene) or alkanes (with Pd/C, full reduction)
- Does NOT reduce isolated C=O under normal conditions (but can reduce C=O with very high pressure)
Catalytic hydrogenation is selective for C=C pi bonds over C=O. If a molecule has both a double bond and a ketone, H₂/Pd reduces the double bond and leaves the ketone alone.
H₂/Pd reduces C=C but not C=O. NaBH₄ reduces C=O but not C=C. This chemoselectivity pair shows up on every exam. Memorize both directions.
One important variant: Lindlar's catalyst 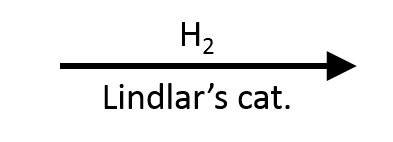 (Pd/CaCO₃/quinoline) is a "poisoned" catalyst that stops alkyne reduction at the cis-alkene stage. If you need a trans-alkene from an alkyne, use Na/NH₃
(Pd/CaCO₃/quinoline) is a "poisoned" catalyst that stops alkyne reduction at the cis-alkene stage. If you need a trans-alkene from an alkyne, use Na/NH₃ 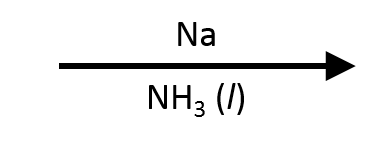 (dissolving metal reduction) instead.
(dissolving metal reduction) instead.
This guide covers the major hydride and catalytic hydrogenation reagents. If your professor also covers Clemmensen reduction (Zn(Hg)/HCl, reduces C=O to CH₂), Wolff-Kishner (H₂NNH₂/KOH/heat, same net result), or Birch reduction (Na/NH₃/ROH, reduces aromatic rings to 1,4-cyclohexadienes), those use different mechanisms but follow the same oxidation state logic. They all move carbon down the ladder.
The Reduction Decision Framework
| I need to convert... | Use this: | Why not the other? |
|---|---|---|
| Aldehyde → 1° alcohol | NaBH₄ or LiAlH₄ | Either works; NaBH₄ is simpler |
| Ketone → 2° alcohol | NaBH₄ or LiAlH₄ | Either works; NaBH₄ is simpler |
| Carboxylic acid → 1° alcohol | LiAlH₄ | NaBH₄ is too weak |
| Ester → 1° alcohol | LiAlH₄ | NaBH₄ is too weak |
| Ester → aldehyde | DIBAL-H (-78°C) | LiAlH₄ goes all the way; NaBH₄ can't start |
| Amide → amine | LiAlH₄ | NaBH₄ is too weak |
| Alkene → alkane | H₂/Pd | Hydride reagents don't reduce C=C |
| Alkyne → cis-alkene | H₂/Lindlar's | Full Pd/C gives alkane; Na/NH₃ gives trans |
| Alkyne → trans-alkene | Na/NH₃ | H₂/Lindlar's gives cis |
| Alkyne → alkane | H₂/Pd (excess) | Lindlar's stops at alkene |
| Ketone + alkene (reduce only alkene) | H₂/Pd | Leaves C=O alone |
| Ketone + ester (reduce only ketone) | NaBH₄ | Leaves ester alone |
Those last two rows are the selectivity problems that separate A students from B students. If your substrate has two reducible groups and the problem asks you to reduce only one, you need to pick the reagent whose reactivity window hits one but not the other.
Quick Check: Your substrate has both an ester and a ketone. You want to reduce only the ketone to an alcohol. Which reagent do you pick?
Reveal answer
NaBH₄. It's strong enough to reduce the ketone but too weak to touch the ester. LiAlH₄ would reduce both. DIBAL-H at -78°C would reduce the ester to an aldehyde and the ketone to an alcohol. Also not what you want. H₂/Pd doesn't reduce either carbonyl. NaBH₄ is the only reagent whose reactivity window covers ketones but not esters.
Oxidation of Alkenes OXIDIZE
The reactions above all involve changes at a carbon that already has an oxygen attached. An alcohol or carbonyl. The next set of reactions is different. Alkene oxidations add new C-O bonds to carbons that had none before.
The oxidation state analysis still applies (you're adding C-O bonds, so it's an oxidation), but the mechanisms are completely different. Alcohol oxidations remove C-H bonds through chromate ester or hydride transfer mechanisms. Alkene oxidations add oxygen across pi systems through cyclic transition states.
These reactions get bundled into the same exam unit because they share the "oxidation" label, not because they work the same way. Don't try to apply the PCC/Jones logic to alkenes, or the mCPBA/OsO₄ logic to alcohols. They're different toolboxes.
Epoxidation: mCPBA (or other peracids) 
Converts C=C to an epoxide (three-membered ring with oxygen). Syn addition. Both C-O bonds form on the same face. The stereochemistry of the alkene is preserved in the product.
mCPBA is the standard reagent. Exam tell: any peracid (RCO₃H) with an alkene = epoxidation.
Syn-Dihydroxylation: OsO₄ (catalytic) / NMO, or cold dilute KMnO₄ 
Adds two -OH groups to the same face of the double bond (syn addition). Gives a 1,2-diol (vicinal diol). OsO₄ with NMO (N-methylmorpholine N-oxide) as the terminal oxidant is the standard method.
Anti-Dihydroxylation: mCPBA then H₃O⁺ (acid-catalyzed epoxide opening)
Adds two -OH groups to opposite faces of the double bond (anti addition). This is a two-step sequence: epoxidize first, then open the epoxide with aqueous acid. The epoxidation is syn, and the ring opening is anti (backside attack), so the net result is anti addition of two -OH groups.
Ozonolysis: O₃ then DMS (or PPh₃) or Zn 
Cleaves the double bond completely. Each carbon of the former C=C gets an oxygen.
- Reductive workup (O₃, then DMS or Zn): gives aldehydes and/or ketones
- Oxidative workup (O₃, then H₂O₂): gives carboxylic acids and/or ketones
Ozonolysis is tested two ways: (1) predict the products from a known alkene, and (2) work backward from ozonolysis products to determine the structure of an unknown alkene. The backward direction is harder and more commonly tested.
Shortcut for ozonolysis products: Put a C=O on each carbon where the double bond was. If that carbon had a hydrogen, it becomes an aldehyde (reductive) or carboxylic acid (oxidative). If it had no hydrogen (disubstituted), it becomes a ketone either way.
Reductive workup (DMS, Zn, PPh₃) = aldehydes survive. Oxidative workup (H₂O₂) pushes those aldehydes to carboxylic acids. Ketones stay ketones either way. Students swap these constantly, don't be one of them.
Some courses test the full molozonide/ozonide mechanism. Others just want the final carbonyl products. Default to the simple "break the double bond and put C=O on each side" approach unless your lecture covered the [2+3] and retro-[2+3] steps.
Oxidative Cleavage: KMnO₄ (hot, acidic, concentrated)
Same net result as ozonolysis with oxidative workup: cleaves the double bond, gives carboxylic acids from disubstituted ends and CO₂ from terminal =CH₂ groups. Less commonly tested than ozonolysis.
Quick Check: You run ozonolysis on 2-butene. With reductive workup (DMS), do you get aldehydes or carboxylic acids?
Reveal answer
Aldehydes. Reductive workup is the gentle one. It preserves the aldehyde products. You'd get two equivalents of acetaldehyde (CH₃CHO). Oxidative workup (H₂O₂) would push those aldehydes further to acetic acid. Remember: reductive = gentler = aldehydes survive.
Predict the products with our Reaction Solver or see full arrow-pushing mechanisms using our Mechanism Solver.
How to Work Through a Problem
Start by identifying the transformation. What functional group are you starting with, and what does the problem want you to end with? Place both on the oxidation state ladder.
If the product is higher on the ladder (fewer C-H bonds, more C-O bonds), you need an oxidizing agent. If it's lower (more C-H bonds), you need a reducing agent. If the carbon skeleton changes between starting material and product, it's not a simple redox problem and needs a different reaction entirely.
Next, ask how far. If you need to go exactly one rung up the ladder (say, primary alcohol to aldehyde), you need a selective reagent like PCC. If you need to go all the way up (primary alcohol to carboxylic acid), you need a strong reagent like Jones. Getting the distance wrong is the most common mistake on oxidation exams: students reach for the first oxidizing agent they remember and overshoot.
Then check for competing functional groups. If the molecule has two reducible groups and the problem only wants one reduced, the reagent has to match the reactivity window: NaBH₄ hits ketones but not esters, H₂/Pd hits alkenes but not carbonyls, LiAlH₄ hits everything. The reference tables above are the lookup; on an exam you reason through it directly.
Three questions (identify the transformation, determine how far, check for selectivity conflicts) are enough to pick the right reagent on most problems.
Quick Check: Convert butanal to 1-butanol. What type of reagent do you need?
Reveal answer
This is a reduction. Place both compounds on the ladder: butanal (aldehyde) sits higher than 1-butanol (alcohol). The target is lower. You're going down the ladder. A surprising number of students reach for an oxidizing agent here because they associate aldehydes with oxidation reactions. The name "butanal" sounds like it should be the product of oxidation, which it is. But the question asks you to go the other direction. NaBH₄ in MeOH handles this cleanly. Always place both compounds on the ladder before you pick a reagent.
Common Exam Patterns
Pattern 1: "Convert 1-butanol to butanal." This is the selective oxidation question. Answer: PCC in CH₂Cl₂. NOT Jones, NOT KMnO₄. Those give butanoic acid.
Pattern 2: "Convert 1-butanol to butanoic acid." Now you want the strong oxidant. Answer: Jones (CrO₃/H₂SO₄/H₂O) or KMnO₄.
Pattern 3: "Reduce methyl benzoate to benzaldehyde." Ester → aldehyde = DIBAL-H at -78°C. If you use LiAlH₄, you get benzyl alcohol (went too far). If you use NaBH₄, nothing happens (too weak for esters).
Pattern 4: "Reduce the ketone without touching the ester." Selective reduction. NaBH₄ reduces ketones but leaves esters alone. LiAlH₄ would reduce both.
Pattern 5: "What happens when a 3° alcohol is treated with PCC?" No reaction. No alpha-hydrogen on the carbinol carbon.
Pattern 6: "Draw the ozonolysis products of 2-methylbut-2-ene." Break the C=C. Put C=O on each side. Left fragment: acetone ((CH₃)₂C=O). Right fragment: acetaldehyde (CH₃CHO) with reductive workup, or acetic acid with oxidative workup.
Worked Examples
Example 1: Multi-Step Oxidation Sequence
Reaction: 1-pentanol → pentanoic acid (one step) AND 1-pentanol → pentanal (one step)
Same starting material, same reaction type, two completely different products. Both transformations go up the oxidation ladder, but by different amounts. Jones goes two steps (alcohol → aldehyde → carboxylic acid); PCC goes one step (alcohol → aldehyde) and stops. The reagent determines where the oxidation halts.
For pentanoic acid, you need Jones reagent (CrO₃, H₂SO₄, H₂O). The aqueous conditions are the key. The aldehyde intermediate hydrates in water, exposing a new C-H bond for a second oxidation. Jones pushes the primary alcohol all the way through to the carboxylic acid.
For pentanal, you need PCC in CH₂Cl₂. The anhydrous solvent is doing the real work. Without water, the aldehyde can't hydrate, so it survives. The oxidation stops at one rung up the ladder.
Answer: Jones → pentanoic acid. PCC → pentanal. This is the single most important distinction in alcohol oxidation, and it comes down to one thing: is there water in the reaction?
Example 2: NaBH₄ vs LiAlH₄ Selectivity
Reaction: Methyl 4-oxopentanoate (ester + ketone in the same molecule)
This molecule has two reducible groups in the same structure: a ketone and an ester. On the oxidation ladder, the ketone sits one rung above the alcohol you want, and the ester sits at the same level as a carboxylic acid, much higher than the ketone. The question is whether you want to hit one or both.
If the problem says "reduce only the ketone," reach for NaBH₄ in MeOH. NaBH₄ delivers hydride to the ketone carbonyl (electrophilic enough to react) but leaves the ester completely untouched. The ester's carbonyl carbon is stabilized by resonance donation from the alkoxy oxygen, which pushes it below NaBH₄'s reactivity threshold. Product: methyl 4-hydroxypentanoate. The ester is still there, intact.
If the problem says "reduce everything," use LiAlH₄ in Et₂O followed by aqueous acid workup. LiAlH₄ is reactive enough to blast through both the ketone and the ester. The ester cleaves to give two alcohols. Product: 1,4-pentanediol, plus methanol lost in the workup.
Answer: NaBH₄ for selective ketone reduction, LiAlH₄ for full reduction. The reagent choice IS the answer on selectivity problems.
Example 3: DIBAL-H, The Ester-to-Aldehyde Conversion
Reaction: Ethyl benzoate + DIBAL-H (1 eq.) at -78°C in toluene
This is the problem that tests whether you know DIBAL-H exists. On the oxidation ladder, esters sit at the same level as carboxylic acids, and the aldehyde target is one rung below.
You need a reagent that goes down exactly one rung from ester and stops. Neither NaBH₄ nor LiAlH₄ can land there.
NaBH₄ won't work. Esters sit below its reactivity threshold, so nothing happens at all. LiAlH₄ is powerful enough to reduce the ester, but it doesn't stop at the aldehyde. It plows straight through to benzyl alcohol. You need a reagent that's strong enough to start the reduction but controlled enough to stop partway. That's DIBAL-H.
At -78°C with one equivalent, DIBAL-H reduces the ester to a tetrahedral aluminum alkoxide intermediate. At that low temperature the intermediate is stable and doesn't collapse further; on aqueous workup, it gives the aldehyde.
Warm the reaction up or use excess DIBAL-H and it keeps going to the alcohol, just like LiAlH₄.
Answer: Benzaldehyde. The -78°C temperature in the problem is the exam signal for DIBAL-H. If you see a low-temperature reduction of an ester, this is what they're testing.
Example 4: Catalytic Hydrogenation Selectivity
Reaction: 4-phenylbut-3-en-2-one + H₂/Pd
This molecule has two potentially reducible sites: a C=C double bond and a C=O ketone. Both are "above" the fully reduced form on the ladder, but they sit on different branches (C=C vs C=O). The question is which branch the reagent operates on.
H₂/Pd works by adsorbing the substrate onto the metal surface and delivering H₂ across the pi bond. It's effective at reducing C=C because alkenes adsorb well onto palladium. But isolated C=O bonds don't adsorb onto the catalyst surface under normal conditions, so the ketone is left alone. The product is 4-phenylbutan-2-one. The double bond is gone, the ketone is still there.
Now flip the reagent. If the same substrate were treated with NaBH₄ instead, the opposite happens: hydride attacks the electrophilic carbonyl carbon (reducing the ketone to an alcohol), but NaBH₄ doesn't touch C=C bonds. You'd get 4-phenylbut-3-en-2-ol. The ketone is gone, the double bond is still there.
Answer: 4-phenylbutan-2-one. H₂/Pd reduces C=C but not C=O. NaBH₄ reduces C=O but not C=C. This reagent pair shows up on exams as a chemoselectivity question. Make sure you can predict both directions.
Example 5: Ozonolysis, Forward Direction
Reaction: 1-methylcyclohexene + O₃, then workup
Ring ozonolysis trips students up because the product doesn't look like what they expect. With an open-chain alkene, ozonolysis gives two separate fragments.
With a cyclic alkene, the double bond is part of the ring, so breaking it doesn't split the molecule in two. Instead, it opens the ring into a single chain with a carbonyl group at each end.
Start by finding the double bond: it sits between C1 and C2 of the cyclohexene ring. Ozone cleaves that bond and puts a C=O on each carbon. C1 had a methyl group attached, so it becomes a ketone. C2 had a hydrogen, so it becomes an aldehyde.
The rest of the ring unfolds into a four-carbon chain connecting the two carbonyls, giving 6-oxoheptanal (a keto-aldehyde).
Answer: With reductive workup (DMS), you get 6-oxoheptanal. With oxidative workup (H₂O₂), the aldehyde end gets pushed further to a carboxylic acid, giving 6-oxoheptanoic acid. The ketone end stays a ketone either way. Ketones can't be oxidized further under these conditions.
A single open-chain dicarbonyl is the correct product for ring ozonolysis, not two separate fragments.
Example 6: Ozonolysis, Backward (Structure Determination)
Reaction: Unknown alkene → ozonolysis with reductive workup (Zn) → acetone + formaldehyde
This is the harder direction and the one that shows up more on exams. You're given the fragments and asked to reconstruct the original alkene.
The logic reverses the forward process. Each ozonolysis product has a C=O that used to be part of the double bond. Strip the oxygen off each carbonyl carbon and reconnect those carbons with a double bond. Acetone gives you (CH₃)₂C= on one side. Formaldehyde gives you =CH₂ on the other. Connect them and you get (CH₃)₂C=CH₂.
Answer: 2-methylpropene (isobutylene). The general method: replace each C=O with C=, then join the two ends. If the ozonolysis gave a single dicarbonyl product instead of two fragments, the original compound was a ring. Connect both C= ends to close it back up.
Example 7: Syn vs Anti Dihydroxylation
Reaction: trans-2-butene with (a) OsO₄/NMO and (b) mCPBA followed by H₃O⁺
Both reactions add two -OH groups across the double bond. The difference is facial selectivity. Which face of the alkene the oxygens end up on.
OsO₄/NMO does syn dihydroxylation. Both -OH groups land on the same face of the former double bond. Starting from trans-2-butene, the two methyl groups are already on opposite faces of the alkene, and that trans relationship is locked in during the syn addition.
When OsO₄ attacks one face of the alkene, both new C-OH bonds form on that same face, giving one specific pair of stereocenters. Attack on the opposite face gives the mirror image. Since both faces are equally likely, you get a 1:1 mixture. The (R,R) and (S,S) enantiomers as a racemic mixture.
The mCPBA/H₃O⁺ sequence does anti dihydroxylation. Epoxidation preserves the trans geometry, then acid-catalyzed ring opening proceeds by backside attack, so the two OHs end up on opposite faces. From trans-2-butene, this gives a molecule with an internal mirror plane: the meso diol, (2R,3S)-2,3-butanediol, achiral despite having two stereocenters.
Answer: Syn gives racemic (±), anti gives meso. Compare this to cis-2-butene (Practice Problem 8), where the results flip: syn gives meso, anti gives racemic. Product stereochemistry depends on both the addition mode AND the starting alkene geometry. "Syn = meso" only works for cis alkenes, so draw the alkene with its substituents in 3D before deciding which stereocenters form.
(a) OsO₄/NMO. Syn Dihydroxylation Mechanism:
(b) mCPBA. Epoxidation Mechanism (first step of anti dihydroxylation):
Example 8: Three Functional Groups, One Reagent
Reaction: A molecule contains an aldehyde, a ketone, and a C=C double bond. You want to reduce only the C=C double bond. Which reagent?
This is the kind of problem that tests whether you actually understand the reagent hierarchy or just memorized a two-column table. Three reducible groups, and you need to hit exactly one.
Start by listing what each reagent does. NaBH₄ reduces aldehydes and ketones but not C=C, which hits two of the three groups you want to leave alone. LiAlH₄ is worse: it reduces the aldehyde, the ketone, and anything else in sight. DIBAL-H at -78°C would reduce the aldehyde and possibly the ketone.
None of the hydride reagents work.
H₂/Pd reduces C=C but doesn't touch C=O under normal conditions. It hits exactly the group you want and leaves the other two alone.
Answer: H₂/Pd. Catalytic hydrogenation is the only common reagent that reduces alkenes while leaving carbonyls intact. The reverse selectivity. Reduce C=O but not C=C. Is handled by NaBH₄. Knowing both directions of this chemoselectivity pair is worth several exam points.
Example 9: Two-Step Synthesis Using Redox
Reaction: Convert 1-pentanol → pentanoic acid → 1-pentanol (round trip, two steps each direction)
Multi-step synthesis problems that involve redox are common on exams. Map the round trip on the ladder first: 1-pentanol (alcohol, rung 2) → pentanoic acid (carboxylic acid, rung 4) is a two-rung climb. Pentanoic acid → 1-pentanol is a two-rung descent. The trick is sequencing reagents correctly and knowing when selectivity matters at each step.
Going up: 1-pentanol to pentanoic acid is a one-step strong oxidation. Use Jones reagent (CrO₃/H₂SO₄/H₂O). You want to blow past the aldehyde, so aqueous conditions are what you need. PCC would stop too early.
Going back down: pentanoic acid to 1-pentanol is a one-step strong reduction. Use LiAlH₄ in Et₂O, then aqueous acid workup. NaBH₄ can't reduce carboxylic acids. Too weak. DIBAL-H at -78°C would stop at the aldehyde stage, which isn't what you want here.
Now consider the more selective version: 1-pentanol → pentanal → 1-pentanol. Step 1 uses PCC (selective, stops at aldehyde). Step 2 uses NaBH₄ (mild, reduces aldehyde back to alcohol). The difference from the round trip above is that every reagent choice must be selective. You can't overshoot in either direction.
Answer: The reagent pairing depends on the target oxidation state. Strong oxidant (Jones) pairs with strong reductant (LiAlH₄) for full transformations. Selective oxidant (PCC) pairs with mild reductant (NaBH₄) for partial transformations. Mixing them up. Using PCC when you need the acid, or LiAlH₄ when you need the aldehyde. Gives the wrong product.
Example 10: Tollens' Test, Distinguishing Aldehydes from Ketones
Reaction: Two unknown liquids (one aldehyde, one ketone) + Tollens' reagent (Ag(NH₃)₂⁺)
This isn't a synthesis problem. It's a detection problem. You have two unknowns and need to tell which is the aldehyde and which is the ketone.
The key difference: aldehydes have a hydrogen on the carbonyl carbon, so they can be oxidized one more step to carboxylic acids. Ketones don't have that hydrogen, so they can't. Tollens' reagent exploits this. The Ag⁺ ion acts as the oxidizing agent. It oxidizes the aldehyde to a carboxylate, and in the process Ag⁺ is reduced to metallic silver, which deposits as a mirror on the inside of the test tube.
The ketone just sits there. No silver mirror forms.
Answer: The aldehyde produces a silver mirror. The ketone gives no reaction. An alternative test is Jones reagent in acetone. The solution turns from orange to green for aldehydes but stays orange for ketones.
Common Exam Mistakes
Mistake 1: Using Jones reagent when the problem asks for an aldehyde Students see "oxidize a primary alcohol" and reach for the first oxidizing agent they remember. Jones reagent in aqueous acid blows past the aldehyde to the carboxylic acid. If the target is an aldehyde, you need PCC, DMP, or Swern.
Mistake 2: Confusing NaBH₄ and LiAlH₄ selectivity NaBH₄ only reduces aldehydes and ketones. LiAlH₄ reduces those plus esters, carboxylic acids, and amides. When a problem has two reducible groups and asks you to hit only one, the reagent choice is the entire answer. Write down what each reagent can and can't reduce before picking.
Mistake 3: Forgetting that 3° alcohols don't oxidize The mechanism requires a C-H bond on the carbinol carbon. Tertiary alcohols don't have one. No oxidizing agent, no matter how strong, changes this. The answer is always "no reaction."
Mistake 4: Getting ozonolysis workup backward Reductive workup (DMS, Zn, PPh₃) → gives aldehydes and ketones. Oxidative workup (H₂O₂) → gives carboxylic acids and ketones. Students frequently swap these. Remember: reductive workup is gentler, so the aldehyde survives.
Mistake 5: Forgetting DIBAL-H exists "Convert an ester to an aldehyde" seems impossible if you only know NaBH₄ and LiAlH₄. NaBH₄ is too weak, LiAlH₄ goes too far. DIBAL-H at -78°C is the answer. If you see ester → aldehyde on an exam, it's testing whether you know this reagent.
Mistake 6: Assuming every "more oxidized product" problem is solved by adding an oxidizing agent Check the carbon skeleton first. Cyclohexanone and cyclohexanecarboxylic acid are both "oxidized," but they have different carbon frameworks. One has C=O inside the ring, the other has COOH hanging off it. Going from one to the other requires C-C bond formation, not a simple redox reaction. If the starting material and product have different numbers of carbons on the key carbon, it's not a redox problem.
Complete Reagent Reference Card
OXIDIZING AGENTS
| Reagent | Tier | Conditions | 1° Alcohol → | 2° Alcohol → | 3° Alcohol → | Notes |
|---|---|---|---|---|---|---|
| PCC | 1 | CH₂Cl₂, RT | Aldehyde | Ketone | NR | Selective; anhydrous |
| Jones | 1 | CrO₃/H₂SO₄/H₂O | Carboxylic acid | Ketone | NR | Strong; aqueous |
| mCPBA | 1 | CH₂Cl₂ | — | — | — | Epoxidizes C=C |
| O₃ then DMS | 1 | CH₂Cl₂, -78°C | — | — | — | Ozonolysis (reductive) |
| O₃ then H₂O₂ | 1 | CH₂Cl₂, -78°C | — | — | — | Ozonolysis (oxidative) |
| DMP | 2 | CH₂Cl₂, RT | Aldehyde | Ketone | NR | Selective; mild; no Cr |
| OsO₄/NMO | 2 | acetone/H₂O | — | — | — | Syn dihydroxylation of C=C |
| Swern | 3 | (COCl)₂/DMSO, -78°C, then Et₃N | Aldehyde | Ketone | NR | Selective; low temp |
| KMnO₄ (conc., hot) | 3 | H₃O⁺ or OH⁻/heat | Carboxylic acid | Ketone | NR | Strong; also cleaves alkenes |
| KMnO₄ (cold, dilute) | 3 | acetone/H₂O, 0°C | — | — | — | Syn-dihydroxylation of C=C (gives 1,2-diol) |
Tier key: 1 = tested on nearly every exam. 2 = commonly tested. 3 = course-dependent. Check your syllabus.
REDUCING AGENTS
| Reagent | Tier | Conditions | Aldehyde → | Ketone → | Ester → | Carboxylic Acid → | C=C → | Notes |
|---|---|---|---|---|---|---|---|---|
| NaBH₄ | 1 | MeOH or EtOH | 1° alcohol | 2° alcohol | NR | NR | NR | Mild; selective |
| LiAlH₄ | 1 | Et₂O or THF, then H₃O⁺ | 1° alcohol | 2° alcohol | 1° alcohol | 1° alcohol | NR | Strong; reduces almost everything |
| H₂/Pd (or Pt) | 1 | EtOAc or EtOH | NR | NR | NR | NR | Alkane | Selective for C=C |
| DIBAL-H | 2 | toluene, -78°C (1 eq.) | 1° alcohol | 2° alcohol | Aldehyde | Aldehyde* | NR | Selective at low T; *carboxylic acids less reliable than esters |
| H₂/Lindlar's | 2 | quinoline | NR | NR | NR | NR | cis-Alkene (from alkyne) | Stops at cis-alkene |
| Na/NH₃ | 2 | liq. NH₃, -33°C | NR | NR | NR | NR | trans-Alkene (from alkyne) | Dissolving metal |
Practice Problem Set
Problems are mixed by type and reagent class. On an exam, you won't get problems sorted by category. Practice the way you'll be tested.
 (in CH₂Cl₂)
→
?
(in CH₂Cl₂)
→
?
Reveal answer
Propanal. PCC in anhydrous CH₂Cl₂ has no water to hydrate the aldehyde, so the oxidation stops one rung up the ladder. Textbook selective 1° alcohol oxidation.
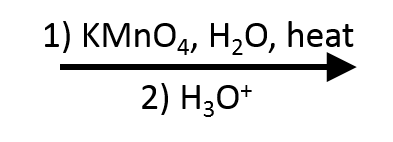


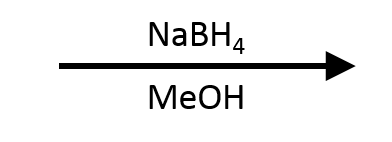
Reveal answer
B) PCC / CH₂Cl₂. Selective oxidant stops at aldehyde. KMnO₄ goes too far (carboxylic acid). The others are reducing agents. Wrong direction entirely.

3. An unknown alkene gives acetone and propanal upon ozonolysis (reductive workup). What is the structure of the alkene?
Reveal answer
2-methylpent-2-ene. Work backward: strip the oxygen from each carbonyl carbon and reconnect with a double bond. Acetone contributes (CH₃)₂C= and propanal contributes =CHCH₂CH₃. Joining them gives (CH₃)₂C=CHCH₂CH₃.
 →
?
→
?
Reveal answer
No reaction. tert-Butanol is a 3° alcohol with no C-H on the carbinol carbon, so every standard oxidation mechanism is blocked. No oxidant, no matter how strong, changes this.

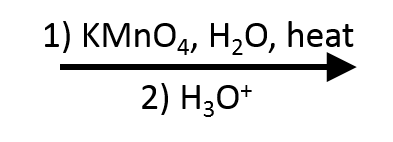
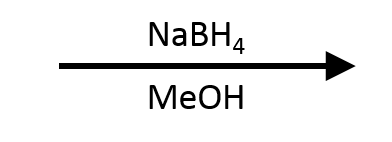

Reveal answer
C) NaBH₄. Reduces the ketone to a 2° alcohol. PCC and KMnO₄ are oxidants. They go the wrong direction. mCPBA is for alkenes, not carbonyls.
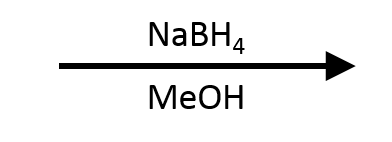
 →
?
→
?
Reveal answer
Propanoic acid. Jones's aqueous conditions let the aldehyde hydrate, exposing a new C-H bond for a second oxidation that pushes all the way to the carboxylic acid. Compare to Problem 1: same substrate, different reagent, completely different product.
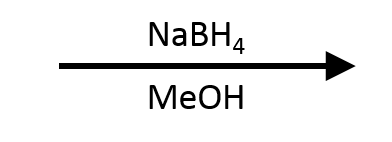

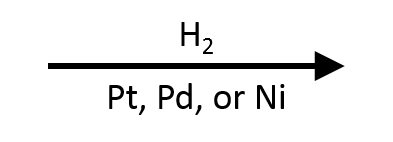
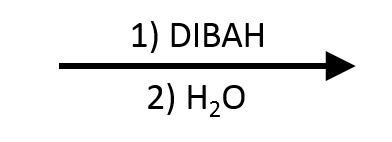
Reveal answer
A) NaBH₄. Reduces the ketone but not the ester. LiAlH₄ reduces both. H₂/Pd doesn't reduce either carbonyl. DIBAL-H at -78°C would reduce the ester to an aldehyde, which isn't what you want.
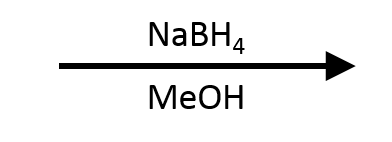
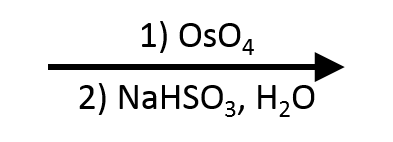 →
?
→
?
Reveal answer
meso-2,3-butanediol. Syn addition from cis-2-butene places both OHs on the same face, giving a molecule with an internal mirror plane (the meso diol). Contrast: syn dihydroxylation of trans-2-butene gives the (R,R)+(S,S) racemate instead, because the methyls are on opposite faces. The product depends on starting geometry, not just the addition mode.
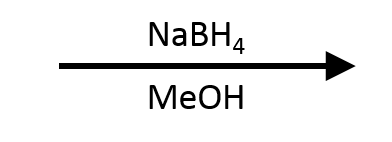

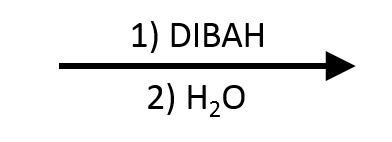
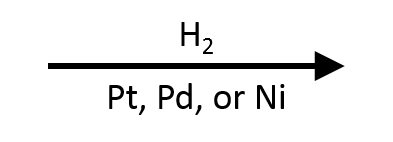
Reveal answer
C) DIBAL-H (-78°C). The only reagent that stops ester reduction at the aldehyde. NaBH₄ can't touch esters at all. LiAlH₄ blows past the aldehyde to the alcohol. H₂/Pd doesn't reduce carbonyls.
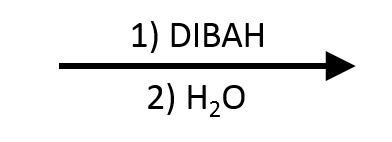
10. A cyclopentene derivative undergoes ozonolysis (reductive workup) to give a single product: pentanedial. What was the starting material?
Reveal answer
Cyclopentene. Ozonolysis of a cycloalkene opens the ring to give a single dicarbonyl product. Pentanedial (OHC-CH₂CH₂CH₂-CHO) has five carbons with an aldehyde at each end. It came from unsubstituted cyclopentene, where ozone broke the only C=C bond.
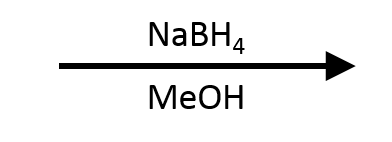 (in MeOH)
→
?
(in MeOH)
→
?
Reveal answer
Benzyl alcohol (C₆H₅CH₂OH). NaBH₄ delivers hydride to the electrophilic carbonyl carbon of the aldehyde, reducing it to a 1° alcohol. The aromatic ring is untouched. NaBH₄ doesn't reduce C=C bonds.



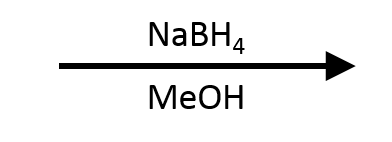
Reveal answer
C) Jones reagent. Strong oxidant goes all the way to carboxylic acid. PCC and DMP both stop at the aldehyde. They're selective oxidants.

 →
?
→
?
Reveal answer
Acetone + acetaldehyde. Ozone cleaves the double bond: the disubstituted end (two methyls) becomes acetone, the monosubstituted end (methyl + H) becomes acetaldehyde. Reductive workup (DMS) preserves the aldehyde without pushing it to a carboxylic acid.
14. Why does Jones reagent oxidize 1° alcohols all the way to carboxylic acids, while PCC stops at the aldehyde?
Reveal answer
Water. Jones reagent uses aqueous conditions. The aldehyde intermediate hydrates in water (forming a geminal diol), which exposes a new C-H bond for a second oxidation. PCC operates in anhydrous CH₂Cl₂. No water means no hydration, so the aldehyde can't be oxidized further.
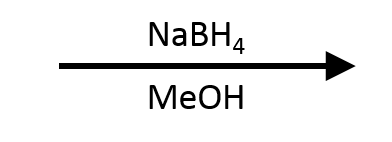 (in MeOH)
→
?
(in MeOH)
→
?
Reveal answer
1-phenylethanol (C₆H₅CH(OH)CH₃). NaBH₄ reduces the ketone to a 2° alcohol. Hydride attacks the carbonyl carbon, giving a secondary alcohol.
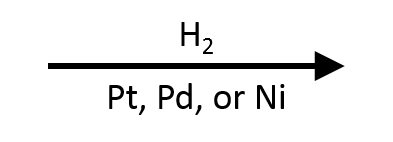
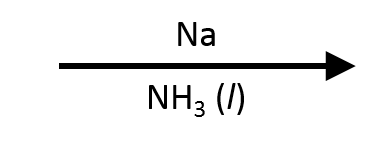

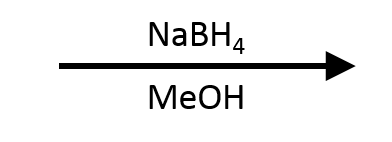
Reveal answer
C) H₂/Lindlar's. The poisoned catalyst stops alkyne reduction at the cis-alkene. H₂/Pd (full catalyst) goes all the way to the alkane. Na/NH₃ gives the trans-alkene instead. NaBH₄ doesn't reduce C≡C bonds.

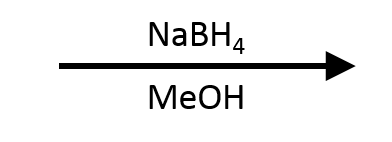 →
?
→
?
Reveal answer
No reaction. The ester oxygen donates into the carbonyl via resonance, dropping the carbonyl carbon's electrophilicity below NaBH₄'s threshold. You'd need LiAlH₄ or DIBAL-H to reduce it.
18. Why can't NaBH₄ reduce esters?
Reveal answer
Resonance stabilization of the ester carbonyl. The ester oxygen donates electron density into the C=O through resonance, making the carbonyl carbon less electrophilic. NaBH₄ is a mild nucleophilic hydride that can only attack sufficiently electrophilic carbons. The ester's reduced electrophilicity puts it below NaBH₄'s reactivity threshold.
 →
?
→
?
Reveal answer
Benzyl alcohol + methanol. LiAlH₄ is strong enough to reduce the ester, which cleaves in two: the acyl side gives benzyl alcohol, and the alkoxy side releases methanol. Two alcohols is the standard outcome for LiAlH₄ + ester.
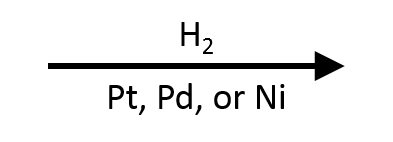
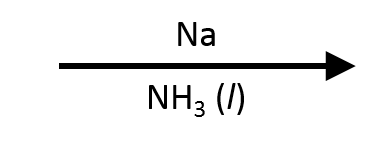


Reveal answer
B) Na/NH₃. Dissolving metal reduction gives the trans-alkene from an alkyne. Lindlar's gives cis. H₂/Pd gives the alkane. LiAlH₄ doesn't reduce C≡C bonds.
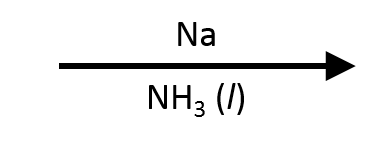
 (in CH₂Cl₂)
→
?
(in CH₂Cl₂)
→
?
Reveal answer
Acetone. PCC oxidizes a 2° alcohol to a ketone. No selectivity issue: ketones can't be oxidized further under these conditions, so PCC and Jones give the same product here.
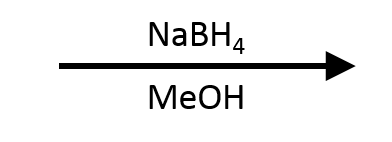

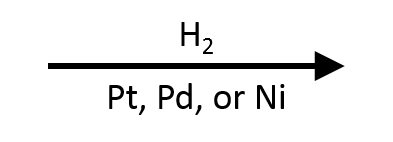
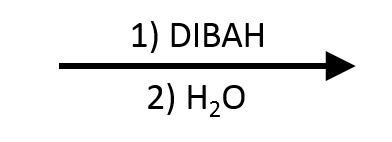
Reveal answer
C) H₂/Pd. Catalytic hydrogenation reduces C=C but leaves C=O alone, which is exactly the selectivity you want. The hydride reagents (NaBH₄, LiAlH₄, DIBAL-H) would all hit the ketone instead of the alkene, even on this α,β-unsaturated enone.
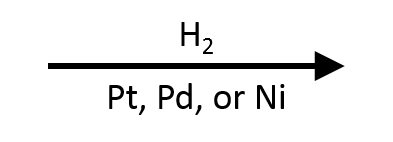
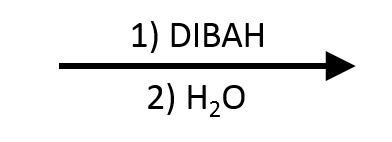 (-78°C, toluene)
→
?
(-78°C, toluene)
→
?
Reveal answer
Benzaldehyde. DIBAL-H at -78°C reduces the ester one step, to the aldehyde, and stops there. The low temperature stabilizes the tetrahedral aluminum alkoxide intermediate so it doesn't collapse further. This is the only way to convert an ester to an aldehyde in one step; the -78°C is the signal.
24. Rank the following carbonyls from easiest to hardest to reduce with NaBH₄: formaldehyde, acetone, methyl acetate, acetic acid.
Reveal answer
Formaldehyde (easiest) > Acetone > Methyl acetate ≈ Acetic acid (hardest / doesn't react). Formaldehyde has the least steric hindrance and the most electrophilic carbonyl. Acetone has more steric bulk from two methyl groups. Esters and carboxylic acids have resonance donation from the oxygen, reducing electrophilicity below NaBH₄'s threshold.
 →
?
→
?
Reveal answer
Cyclohexene oxide (1,2-epoxycyclohexane). mCPBA transfers an oxygen to the double bond in a syn fashion, forming the three-membered epoxide ring. The stereochemistry of the alkene is preserved in the product.
26. An unknown diene undergoes ozonolysis (reductive workup) and gives three carbonyl products: formaldehyde, glyoxal (OHC-CHO), and acetone. Determine the structure of the diene.
Reveal answer
4-methylpenta-1,3-diene, CH₂=CH-CH=C(CH₃)₂. Work backward: formaldehyde (=CH₂) is one terminus, acetone (=C(CH₃)₂) is the other, and glyoxal comes from the two internal carbons that each formed a C=O when their adjacent double bonds were cleaved. Connect them as CH₂=CH-CH=C(CH₃)₂. Don't confuse this with isoprene (2-methylbuta-1,3-diene, CH₂=C(CH₃)-CH=CH₂), which has only five carbons and gives 2× formaldehyde + methylglyoxal instead.
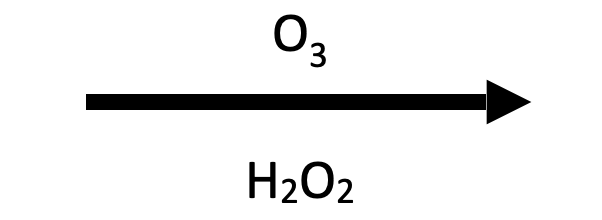 →
?
→
?
Reveal answer
Acetone + acetic acid. Same cleavage pattern as reductive ozonolysis, but oxidative workup (H₂O₂) pushes the aldehyde fragment further to a carboxylic acid. The ketone fragment (acetone) stays the same either way. Ketones can't be oxidized further.
28. A student treats a 3° alcohol with excess KMnO₄ under heating. What is the product?
Reveal answer
No reaction. 3° alcohols cannot be oxidized, regardless of how strong the oxidant is or how much you heat it. The mechanism requires removal of a C-H bond from the carbinol carbon, and there isn't one.
Oxidation and Reduction in Organic Chemistry is only available for OrgoSolver Pro members
Get unlimited access to our content
Interactive study tools like the Mechanism Solver or Resonance Solver, Study Guides, Lab Reports, and more!