SN1 vs SN2 vs E1 vs E2: How to Choose the Right Mechanism
The four-question system for picking the right mechanism
SN1 vs SN2 vs E1 vs E2: How to Choose the Right Mechanism
If you're taking organic chemistry, this is probably the topic keeping you up at night. An exam gives you a substrate, a nucleophile/base, and a solvent, and you're supposed to predict whether the reaction runs SN1, SN2, E1, or E2. Sometimes it's a mix. Sometimes there's no reaction at all.
There are four things to check, in order: substrate, nucleophile/base, solvent, and temperature. Work through them systematically and the answer is usually unambiguous. This guide covers what each reaction does, then runs worked examples using OrgoSolver's mechanism solver so you can see the electron-pushing diagrams.
The Four Reactions at a Glance
Before diving into how to choose, here's what each reaction actually does:
Substitution reactions replace the leaving group with a nucleophile. The carbon skeleton stays intact. Only the group attached to the electrophilic carbon changes.
Elimination reactions remove the leaving group and a proton from an adjacent carbon, forming a new pi bond (usually a double bond). You lose two groups and gain unsaturation.
| SN1 | SN2 | E1 | E2 | |
|---|---|---|---|---|
| What happens | Nucleophile replaces leaving group | Nucleophile replaces leaving group | Base removes H, leaving group departs, double bond forms | Base removes H, leaving group departs, double bond forms |
| Steps | 2 (carbocation intermediate) | 1 (concerted, no intermediate) | 2 (carbocation intermediate) | 1 (concerted, no intermediate) |
| Stereochemistry | Racemization (mix of R and S) | Inversion (backside attack) | No stereospecificity | Anti-periplanar required |
| Rate law | Rate = k[substrate] | Rate = k[substrate][nucleophile] | Rate = k[substrate] | Rate = k[substrate][base] |
How to Decide: Four Factors
Factor 1: The Substrate
Start here. The structure of the carbon bearing the leaving group matters more than anything else. Count the carbons attached to the electrophilic carbon.
Methyl (CH₃-LG) and Primary (1°): SN2 is strongly favored because the electrophilic carbon is unhindered, giving the nucleophile easy backside access. SN1 and E1 don't happen here because methyl and primary carbocations are too unstable to form. The one exception is E2, and it only shows up with a strong, bulky base like t-BuOK. If the base is too bulky to attack carbon, it grabs a proton instead and you get elimination. Without a bulky base, primary substrates default to SN2 almost every time.
Secondary (2°): All four reactions are possible here, which is why professors love putting secondary substrates on exams. The nucleophile/base, solvent, and temperature are what break the tie.
Tertiary (3°): SN2 does not happen at tertiary carbons, period. Three bulky substituents block backside attack completely, which leaves SN1, E1, and E2.
With a weak nucleophile in a polar protic solvent, SN1 dominates. With a strong base, E2 takes over. E1 shows up with a polar protic solvent and heat but no strong base.
If you see a tertiary substrate, immediately cross SN2 off your list. If you see methyl or primary, cross SN1 and E1 off. This alone eliminates half the possibilities before you even look at the reagent.
Substrate tells you what's off the table. Tertiary → no SN2. Methyl or primary → no SN1, no E1. Decide this before you look at the reagent.
Quick Check: A secondary substrate shows up on your exam. Which mechanisms can you immediately rule out?
Reveal answer
None. All four are possible at secondary carbons, which is precisely why secondary substrates dominate exams. You need the other three factors to decide.
Factor 2: The Nucleophile/Base
The same species can act as a nucleophile (attacks carbon) or a base (attacks hydrogen). What it does depends on its properties:
Strong nucleophile + weak base → SN2 Examples: CN⁻, I⁻, RS⁻, N₃⁻ These species are good at attacking carbon but don't readily grab protons. They drive substitution.
Strong base + strong nucleophile → SN2 or E2 (competing) Examples: OH⁻, RO⁻ (like CH₃O⁻, EtO⁻) These can do either substitution or elimination. Substrate structure breaks the tie: primary substrate → SN2, tertiary → E2.
Strong, bulky base → E2 Examples: t-BuOK (potassium tert-butoxide), LDA, DBU The bulk prevents the base from attacking carbon (too crowded for SN2), so it attacks the more accessible proton instead. Bulky base = elimination.
Weak nucleophile/weak base → SN1 or E1 Examples: H₂O, ROH (like CH₃OH, EtOH) These aren't reactive enough to force a concerted mechanism. They wait for the substrate to ionize on its own (forming a carbocation), then react. This is why SN1/E1 require substrates that form stable carbocations (secondary or tertiary).
Quick Check: You see NaOCH₃ as the reagent. Is this a strong nucleophile, strong base, or both? What mechanism(s) does it favor?
Reveal answer
Both. Methoxide (CH₃O⁻) is a strong nucleophile and a strong base, but it's not bulky. It favors SN2 with primary substrates and E2 with tertiary substrates. With secondary substrates, you'll likely get a mixture.
Factor 3: The Solvent
Polar protic solvents (water, alcohols, acetic acid):
- Stabilize carbocations through solvation → favor SN1 and E1
- Solvate and weaken nucleophiles → disfavor SN2
Polar aprotic solvents (DMSO, DMF, acetone, acetonitrile):
- Don't stabilize carbocations well → disfavor SN1 and E1
- Don't solvate nucleophiles → nucleophiles remain strong → favor SN2
Polar protic → SN1/E1 territory. Polar aprotic → SN2 territory.
Quick Check: A reaction is run in ethanol. Does this help or hurt SN2? Why?
Reveal answer
It hurts SN2. Ethanol is polar protic. It solvates and weakens nucleophiles through hydrogen bonding, slowing backside attack. Polar protic solvents push toward SN1/E1 instead. A strong nucleophile in a polar protic solvent is weaker than you'd expect, so solvent can override what the reagent alone suggests.
Factor 4: Temperature
Heat favors elimination over substitution. Elimination creates two molecules from one (alkene + HX), so entropy is on its side at higher temperatures. The higher activation energy of elimination also means it benefits more from added heat (Arrhenius).
If a problem says "heat" or "Δ," shift your prediction toward E1 or E2. No temperature mentioned? Default to substitution.
Heat tips the answer from substitution to elimination. SN1 becomes E1, SN2 becomes E2. No heat mentioned, no reason to elim-shift.
Quick Check: A tertiary substrate reacts with a weak nucleophile in a polar protic solvent. You predict SN1. Now the problem adds "heat." Does your answer change?
Reveal answer
Yes. Heat shifts the competition toward elimination. The answer becomes E1 (or a mixture of SN1 and E1, with E1 as the major product). Temperature is the tiebreaker between substitution and elimination when all other factors point to unimolecular mechanisms.
Special Case: Alcohol Substrates (Acid-Catalyzed Dehydration)
Everything above assumes an alkyl halide or tosylate as the substrate. Alcohols come up just as often on exams, and the rules shift slightly.
The problem with alcohols: -OH is a terrible leaving group. Hydroxide is too basic and too small to depart on its own. So none of the four mechanisms (SN1, SN2, E1, E2) happen with an alcohol under normal conditions.
The fix: protonate it first. Treating an alcohol with a strong acid (H₂SO₄, H₃PO₄, or TsOH) converts -OH into -OH₂⁺, which is water, an excellent leaving group. Once protonated, the substrate behaves like any other good-leaving-group system and the same four-factor analysis applies.
Acid-catalyzed dehydration is the most common exam scenario for alcohol substrates. The general reaction: alcohol + strong acid + heat → alkene + water. This proceeds by E1:
- Protonation: Acid donates H⁺ to the -OH, converting it to -OH₂⁺
- Loss of water: The -OH₂⁺ departs to form a carbocation (this is the rate-determining step)
- Deprotonation: A base (often the conjugate base of the acid, or another alcohol molecule) removes a β-hydrogen, forming the alkene
Because a carbocation intermediate forms, all the rules of E1 apply: Zaitsev's rule gives the major product (most substituted alkene), carbocation rearrangements (1,2-hydride and 1,2-methyl shifts) are possible, and the order of reactivity is 3° > 2° > 1° (matching carbocation stability).
Whenever you see alcohol + H₂SO₄ (or H₃PO₄) + heat on an exam, that's E1 dehydration. No nucleophile present, no strong base, just acid and heat pushing toward elimination.
If you want a quick-reference version of the four factors above, the Decision Cheat Sheet table in the gated section covers every substrate/reagent/solvent combination in one scannable grid.
How to Work Through a Problem
Start with the substrate. Count the carbons on the electrophilic carbon and immediately eliminate what cannot happen. Methyl or primary? Cross off SN1 and E1. Tertiary? Cross off SN2. You have just cut your options in half before reading the rest of the problem.
Next, look at the reagent. Classify it as strong nucleophile, strong base, bulky base, or weak.
If it's a strong nucleophile that's a weak base (CN⁻, I⁻), substitution wins. If it's a strong bulky base (t-BuOK), elimination wins. If it's weak (water, an alcohol), you need a stable carbocation, so SN1/E1 territory.
If the reagent is both a strong nucleophile and a strong base (OH⁻, RO⁻), the substrate breaks the tie: primary favors SN2, tertiary favors E2, secondary gives a mixture.
Then check the solvent. Polar protic solvents stabilize carbocations and weaken nucleophiles, pushing toward SN1/E1. Polar aprotic solvents keep nucleophiles strong, pushing toward SN2.
Finally, check for heat. If the problem says "heat" or shows Δ, shift your answer toward elimination (E1 or E2). No temperature mentioned? Default to substitution.
That is the entire decision process: substrate → reagent → solvent → temperature. Run through it in order and the answer falls out. The worked examples below walk through this sequence on real reactions so you can see it in action.
Worked Examples
Example 1: Clear-cut SN2
Reaction: 1-bromobutane + NaCN in DMSO
Start with the substrate. 1-bromobutane is primary, so the bromine is on a carbon attached to only one other carbon. That immediately rules out SN1 and E1, since a primary carbocation would be required and those are too unstable.
Now the nucleophile. Cyanide (CN⁻) is one of the best nucleophiles in organic chemistry, but it's a surprisingly weak base (the pKa of HCN is only about 9). That combination, strong nucleophile and weak base, points firmly toward SN2 over E2.
The solvent confirms it. DMSO is polar aprotic, which means it won't solvate the cyanide and weaken it. The nucleophile stays fully reactive.
Answer: SN2. The product is pentanenitrile, formed by backside attack with inversion of configuration.
Example 2: Clear-cut E2
Reaction: 2-bromo-2-methylpropane + potassium tert-butoxide (t-BuOK) in t-BuOH
The substrate is tert-butyl bromide, tertiary. Write down "no SN2" before you do anything else. That eliminates one of four possibilities immediately.
The reagent is potassium tert-butoxide, which is both a strong base and extremely bulky (three methyl groups surrounding the oxygen). The steric bulk makes it impossible for this base to approach the electrophilic carbon for substitution. It can only reach the smaller, more accessible β-hydrogens, which means elimination.
Even if t-BuOK weren't bulky, a strong base with a tertiary substrate gives E2. The bulk just removes any remaining ambiguity.
Answer: E2. The product is 2-methylpropene. Only one possible alkene here since all the β-hydrogens are equivalent.
Example 3: Clear-cut SN1
Reaction: 2-bromo-2-methylpropane + water
Tertiary substrate, so cross SN2 off immediately. Water is both the nucleophile and the solvent here, and it's weak and polar protic, which is textbook SN1 territory. No strong base, no heat, so the carbocation just gets captured by water.
Answer: SN1
Try working through these mechanisms yourself using our Mechanism Solver or predict the products with our Reaction Solver.
More Worked Examples
Example 4: The Tricky Secondary Substrate
Reaction: 2-bromopentane + NaOCH₃ in methanol
The substrate is secondary, so all four mechanisms are on the table. That means the reagent has to break the tie. Sodium methoxide gives you CH₃O⁻, which is both a strong nucleophile and a strong base. And it's not bulky.
The solvent is methanol, polar protic, which normally pushes toward SN1/E1 territory. But the strong base overrides the solvent effect here: CH₃O⁻ is reactive enough to force a concerted mechanism rather than waiting for the substrate to ionize. No heat is mentioned, so there's no extra push toward elimination.
Answer: SN2 and E2 competing. You'll get a mixture of substitution and elimination products. This setup does not have a single clean answer, and any exam that expects one is oversimplifying. At room temperature, SN2 is slightly favored because CH₃O⁻ is a good nucleophile and the secondary substrate still allows backside attack, so if forced to pick, lead with the substitution product. Write both products if you have space.
But if the question specifies heat, E2 becomes the major pathway.
Textbook vs exam reality: most exams expect a single clean answer for secondary + strong base problems, but in practice you get a mixture. At room temperature SN2 is slightly favored; with heat, E2 wins. Write both products when the question allows it.
Example 5: SN1 with Carbocation Rearrangement
Reaction: 3-bromo-2-methylbutane + ethanol
This one catches people. Secondary substrate, weak nucleophile, polar protic solvent. You correctly identify SN1. You draw the carbocation. You write the product. And you lose points.
Why? Because the initial secondary carbocation isn't the final carbocation. A hydrogen on the adjacent carbon can shift over (1,2-hydride shift) to give a more stable tertiary carbocation. The nucleophile attacks that carbocation, not the one you drew first.
This is the kind of problem that separates B students from A students on the second exam. You can know the whole mechanism and still lose points if you don't check for rearrangements before you write the product.
Answer: SN1 with rearrangement
Example 6: E2 Regiochemistry (Zaitsev vs Hofmann)
Reaction: 2-bromo-2-methylbutane + NaOEt
Analysis: Tertiary substrate + strong base = E2. But WHERE does the double bond form?
With a non-bulky base like EtO⁻, the Zaitsev product (more substituted alkene) is major: 2-methylbut-2-ene.
If we switch to t-BuOK, the Hofmann product (less substituted alkene) is major: 2-methylbut-1-ene (CH₂=C(CH₃)CH₂CH₃). The bulky base abstracts the more accessible (less hindered) proton on the methyl group at C1.
Example 7: No Reaction
Reaction: Neopentyl bromide (2,2-dimethyl-1-bromopropane) + NaOH in DMSO
Analysis:
- Substrate: Primary, but VERY bulky (the carbon next to the leaving group has three methyl groups)
- SN2? Technically primary, but the steric hindrance is enormous. Blocked in practice
- SN1? Primary carbocation would be required, too unstable
- E2? NaOH is a strong base, but the β-carbon (the quaternary C next to CH₂Br) has zero hydrogens. With no β-H available, E2 cannot happen at all
- E1? Requires carbocation, too unstable
Answer: Very slow SN2 at best, no reaction under normal conditions. The trap here is seeing "primary" and defaulting to SN2 without checking steric environment.
Example 8: Allylic/Benzylic Substrates (Special Cases)
Reaction: Benzyl bromide (C₆H₅CH₂Br) + H₂O
The substrate looks primary. There's only one carbon between the ring and the leaving group. Normally, primary + weak nucleophile (water) means nothing useful happens. Primary carbocations are too unstable for SN1, and water is too weak to force SN2 on its own.
But this is a benzylic position. The carbocation that forms when bromide leaves is stabilized by resonance with the aromatic ring. A primary benzylic carbocation is roughly as stable as a regular secondary carbocation, which is stable enough to form. Water then attacks the carbocation as the nucleophile.
Answer: SN1. The benzylic carbocation is resonance-stabilized, making ionization feasible despite the primary classification. The same logic applies to allylic substrates. Any time the carbocation can delocalize into a pi system, SN1 becomes possible even at positions where you'd normally rule it out.
Example 9: E2 and the Anti-Periplanar Requirement
Reaction: trans-1-bromo-4-tert-butylcyclohexane + NaOEt
Analysis: This tests whether students understand E2's geometric requirement. E2 requires the H and leaving group to be anti-periplanar (180° dihedral angle). In cyclohexane chairs, this means both must be axial.
The tert-butyl group locks the ring (always equatorial due to its size). For the trans isomer specified here, with t-Bu equatorial at C4, Br sits axial at C1 in the preferred chair. This means the β-hydrogens at C2 and C6 that are also axial are anti-periplanar to Br, so E2 proceeds readily.
The cis isomer is where the geometry problem arises: t-Bu equatorial forces Br equatorial as well, removing anti-periplanar geometry and making E2 extremely slow. That's the tricky case, but for the trans isomer given here, the geometry works in our favor.
Answer: E2 proceeds. For the trans isomer, Br is axial when t-Bu is equatorial → anti-periplanar H atoms are available → E2 proceeds. The cis isomer is where the geometry problem arises. Always draw the chair to confirm.
E2 on a cyclohexane needs an axial leaving group with an axial β-H next to it. Both substituents must be axial for the anti-periplanar geometry E2 requires. No axial β-H next to the axial leaving group, no E2.
Example 10: Vinyl and Aryl Substrates
Reaction: Bromobenzene + NaOH
Analysis:
- Substrate: Aryl (sp² carbon bears the leaving group)
- SN2? Impossible. Backside attack on an sp² carbon in a ring can't occur
- SN1? Phenyl cation is extremely unstable
- E1/E2? No β-hydrogens in the traditional sense
Answer: No reaction under normal SN/E conditions. Aryl halides need special conditions (SNAr with electron-withdrawing groups, or benzyne mechanisms with very strong bases like NaNH₂). Don't confuse aryl halides for regular sp³ substrates.
Example 11: Alcohol Dehydration (E1)
Reaction: 2-methylcyclohexanol + H₂SO₄, heat
The substrate is an alcohol, and -OH by itself is a terrible leaving group. But sulfuric acid fixes that. It protonates the hydroxyl to -OH₂⁺, turning it into water, which is an excellent leaving group. There's no nucleophile present, just acid and heat. Heat always pushes toward elimination, and with no strong base around to force a concerted E2, this goes through E1. The secondary carbon loses water to form a 2° carbocation, and then a β-hydrogen is removed to give the alkene.
After the carbocation forms, deprotonation of a β-hydrogen gives the alkene. Zaitsev's rule applies: the more substituted double bond is the major product. Here that's 1-methylcyclohexene (trisubstituted alkene), not 3-methylcyclohexene or methylenecyclohexane.
Answer: E1 dehydration → 1-methylcyclohexene (Zaitsev product, major) + 3-methylcyclohexene (minor)
Example 12: Alcohol Dehydration with Rearrangement
Reaction: 3,3-dimethyl-2-butanol + H₂SO₄, heat
Alcohol + acid + heat, so you know it's E1 dehydration. Protonate the -OH, lose water, and you've got a 2° carbocation at C2. Most students stop here, deprotonate, and write 3,3-dimethylbut-1-ene.
But look at C3. It has three methyl groups sitting right next to that electron-deficient C2. One of those methyls can migrate (1,2-methyl shift), and now you have a 3° carbocation instead of a 2°. The 3° carbocation is more stable, and when you deprotonate it, you get 2,3-dimethylbut-2-ene, a tetrasubstituted alkene.
The rearranged product wins twice: the carbocation is more stable AND the resulting alkene is more substituted. The unrearranged product (3,3-dimethylbut-1-ene) is minor.
Answer: E1 with rearrangement → 2,3-dimethylbut-2-ene (major) + 3,3-dimethylbut-1-ene (minor)
Any time you see an alcohol dehydration, form the carbocation first, then ask: could a hydride or methyl shift create a more stable carbocation? If yes, the rearranged product is usually major.
Example 13: E2 Stereochemistry (Newman Projection Analysis)
Reaction: (R)-2-bromobutane + NaOEt
Secondary substrate + strong base = E2. That part is easy. The hard part is predicting the stereochemistry of the product.
Draw the Newman projection first. Sight down C2→C3, place Br on C2, and find which H on C3 is anti (180°) to the leaving group. That's the H that gets removed.
For (R)-2-bromobutane, the H that's anti to Br leads to the trans alkene (E-but-2-ene). The other β-H is gauche to Br, which would give cis-but-2-ene, but the anti pathway dominates because E2 is stereospecific.
Answer: trans-but-2-ene (E-but-2-ene), major. It's both Zaitsev AND anti-periplanar favored. The cis isomer is minor.
E2 from open-chain substrates almost always gives the trans (E) alkene. The anti-periplanar geometry naturally places the largest groups on opposite sides of the forming double bond.
E2 + open chain = trans (E) alkene, Zaitsev. Anti-periplanar geometry forces the largest groups onto opposite sides of the forming double bond. The cis isomer is minor.
Common Exam Mistakes
Writing SN2 for a tertiary substrate. Tertiary + SN2 = zero credit. It doesn't matter what nucleophile or solvent you have.
Assuming primary always means SN2. Usually yes, but check for neopentyl-type steric hindrance or resonance-stabilized carbocations (allylic, benzylic) that could enable SN1.
Ignoring solvent. We've seen students lose points because they wrote "SN2" for a primary substrate just because the nucleophile was strong, without noticing the solvent was water. A polar protic solvent solvates and weakens the nucleophile. CN⁻ in DMSO is a completely different beast than CN⁻ in water. Always check the solvent.
Forgetting carbocation rearrangements in SN1/E1. Whenever a carbocation forms, check whether a 1,2-hydride shift or 1,2-methyl shift could create a more stable carbocation. Professors love testing this.
Not drawing the chair for E2 in cyclic systems. In open-chain substrates, bond rotation makes anti-periplanar geometry accessible for any β-hydrogen, so you can skip the conformational analysis. Rings are different. In a cyclohexane chair, the leaving group must be axial, and the β-hydrogen being removed must also be axial on the same side of the ring. Picture a locked cyclohexane chair where the leaving group is equatorial: no adjacent H is anti-periplanar, and E2 becomes extremely slow. If you're not drawing the chair on E2 problems with rings, you're guessing.
Forgetting rearrangements in alcohol dehydrations. Students correctly identify E1, form the carbocation, then forget to check for shifts before writing the product. The rearranged alkene is usually the major product.
Predicting cis alkenes from E2 when trans is favored. Draw the Newman projection. The anti arrangement of H and leaving group naturally places the largest groups on opposite sides of the double bond, giving the trans (E) alkene as the major product. The cis (Z) isomer requires a gauche arrangement and is minor.
Quick Reference: Decision Cheat Sheet
| Scenario | Substrate | Nuc/Base | Solvent | Temp | → Reaction |
|---|---|---|---|---|---|
| 1 | Methyl/1° | Strong nuc | Polar aprotic | RT | SN2 |
| 2 | Methyl/1° | Strong bulky base | Any | Any | E2 |
| 3 | Methyl/1° | Weak nuc | Polar protic | Any | No rxn |
| 4 | 2° | Strong nuc | Polar aprotic | RT | SN2 |
| 5 | 2° | Strong base | Any | Heat | E2 |
| 6 | 2° | Strong base | Any | RT | SN2 / E2 mix |
| 7 | 2° | Weak nuc | Polar protic | RT | SN1 |
| 8 | 2° | Weak nuc | Polar protic | Heat | SN1 / E1 |
| 9 | 3° | Strong base | Any | Any | E2 |
| 10 | 3° | Weak nuc | Polar protic | RT | SN1 |
| 11 | 3° | Weak nuc | Polar protic | Heat | E1 (+ SN1) |
| 12 | 3° | Strong nuc | Polar aprotic | Any | E2 (not SN2!) |
| 13 | 3° alcohol | H₂SO₄ (acid) | — | Heat | E1 dehydration |
| 14 | 2° alcohol | H₂SO₄ (acid) | — | Heat | E1 dehydration |
| 15 | 1° alcohol | H₂SO₄ (acid) | — | Heat | E2 dehydration (neopentyl-type primaries without a β-H rearrange to a 3° cation first, then lose a proton by E1) |
| 16 | Alcohol | HBr or HI | — | RT or Heat | SN1 or SN2 (substitution, not elimination) |
Practice Problems
Test your understanding. For each reaction, determine the major pathway: SN1, SN2, E1, E2, or No Reaction.
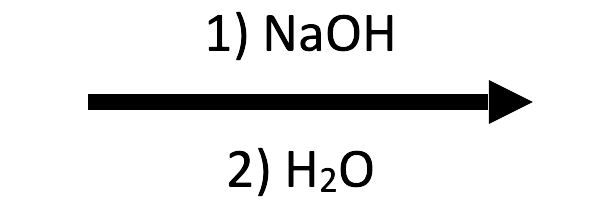 →
?
→
?
Reveal answer
E2 major, SN1 competing. Tertiary rules out SN2; hydroxide drives E2 while water (polar protic) stabilizes the tert-butyl cation enough for SN1 to run alongside. Expect 2-methylpropene as the major product with some tert-butyl alcohol from SN1.
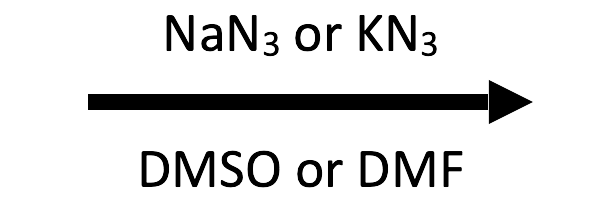 →
?
→
?
Reveal answer
SN2. Azide is a strong nucleophile but a weak base, so it attacks carbon instead of driving elimination. DMSO keeps it unsolvated and fully reactive against the secondary substrate.
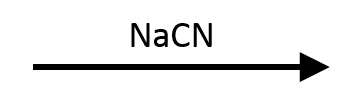 (in DMSO)
→
?
(in DMSO)
→
?
Reveal answer
SN1/E1, with some E2. Tertiary rules out SN2, but CN⁻ is only a weak base (pKa of HCN ≈ 9), so it won't cleanly force E2 either. Expect a slow mixture: SN1 gives tert-butyl cyanide, E1 gives 2-methylpropene, and a minor E2 pathway runs alongside. No single mechanism dominates when the base is weak.
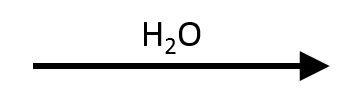 →
?
→
?
Reveal answer
SN1. Water is both the nucleophile and the solvent, and it's weak on both counts, which rules out SN2 and E2. The polar protic environment stabilizes the secondary carbocation, which water then captures to give 2-butanol.
 →
?
→
?
Reveal answer
SN2. Primary + weak base rules out SN1/E1/E2 by default. Iodide is a strong, polarizable nucleophile, and polar aprotic acetone keeps it fully reactive at the electrophilic carbon.
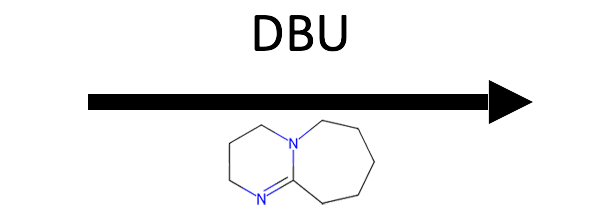 →
?
→
?
Reveal answer
E2. Tertiary rules out SN2. DBU is a strong base but non-nucleophilic (its lone pairs are sterically shielded), so it abstracts a proton instead of attacking carbon. Tertiary + strong base = E2.
 (in DMSO)
→
?
(in DMSO)
→
?
Reveal answer
SN2. Cyanide is a strong nucleophile but a weak base (pKa of HCN ≈ 9), so substitution wins over elimination at the secondary carbon. DMSO keeps it unsolvated and fully reactive.
 →
?
→
?
Reveal answer
No reaction. Neopentyl bromide is technically primary, but the adjacent quaternary carbon blocks SN2 sterically and removes all β-hydrogens, killing E2. A primary cation rules out SN1/E1. All four pathways are closed.
 →
?
→
?
Reveal answer
E2, but geometry matters. The substrate is secondary (the carbon bearing Br has two ring CH₂ neighbors and one H), and t-BuOK's bulk makes E2 the right pathway. In the trans isomer shown, the locked-equatorial t-Bu forces Br axial, placing an adjacent axial H anti-periplanar for E2 to proceed. In the cis isomer Br would be equatorial and no axial β-H would be available, shutting E2 down.
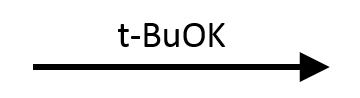 →
?
→
?
Reveal answer
E2. t-BuOK is strong but too bulky for backside attack, so it grabs an accessible β-hydrogen instead. Bulky base + secondary substrate is one of the cleanest E2 setups on an exam.
 →
?
→
?
Reveal answer
No reaction. The C-Br is on an sp² aromatic carbon, so SN2 has no back side, SN1 would need a catastrophically unstable phenyl cation, and E1/E2 have no β-hydrogens to abstract. Aryl halides need special mechanisms (SNAr, benzyne) that aren't set up here.
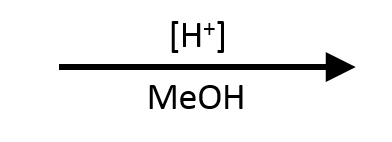 →
?
→
?
Reveal answer
SN1. Tertiary rules out SN2, and methanol is a weak nucleophile/base in its own polar protic solvent, which is classic SN1 conditions. The tert-butyl cation forms, methanol captures it, and the product is tert-butyl methyl ether.
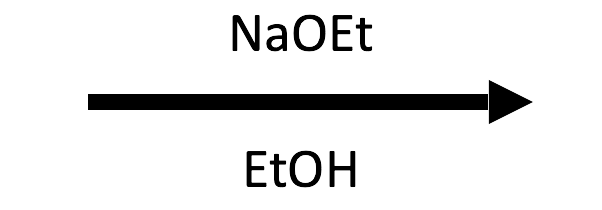 (80°C)
→
?
(80°C)
→
?
Reveal answer
E2. Sodium ethoxide at a secondary substrate would normally give an SN2/E2 mix at room temperature, but 80°C tips the balance to elimination: E2 has a higher activation energy than SN2 (so it benefits more from heat) and produces two molecules from one (entropy favors it).
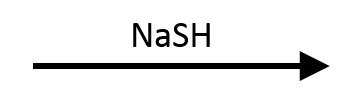 (in DMF)
→
?
(in DMF)
→
?
Reveal answer
SN2. A methyl substrate plus a polarizable nucleophile (sulfur's diffuse electron cloud overlaps easily with the electrophilic carbon) plus polar aprotic DMF is about as clean as SN2 gets.
 →
?
→
?
Reveal answer
SN2. Primary + non-bulky strong nucleophile defaults to substitution via backside attack, giving the ether (Williamson synthesis).
 →
?
→
?
Reveal answer
E2. Primary normally means SN2, but t-BuOK's three methyl groups block backside attack, so it abstracts a β-hydrogen instead. This is the one common scenario where a primary substrate gives elimination.
 →
?
→
?
Reveal answer
SN2 with inversion. Iodide is a strong nucleophile but a very weak base, and polar aprotic acetone keeps it reactive at the secondary carbon. Backside attack inverts the stereocenter, so (R)-2-bromobutane becomes (S)-2-iodobutane.
Alcohol Dehydration Problems:
Reveal answer
E1 dehydration → cyclohexene. Acid protonates -OH to -OH₂⁺, water departs forming 2° carbocation, deprotonation gives cyclohexene. Only one possible alkene product here.
Reveal answer
E1 dehydration → 2-methylbut-2-ene (Zaitsev). Secondary alcohol, acid + heat → E1. Carbocation at C2 is 2°, no rearrangement needed (shifting wouldn't improve stability). Deprotonation follows Zaitsev's rule: more substituted alkene (trisubstituted) is major over 3-methylbut-1-ene (monosubstituted).
Reveal answer
E1 with rearrangement → 2-methylbut-2-ene. This is neopentyl alcohol. After protonation and water loss, a 1° carbocation forms, but 1° carbocations are unstable, so a 1,2-methyl shift occurs immediately to give a 3° carbocation. Deprotonation of the rearranged cation gives 2-methylbut-2-ene. Students who miss the rearrangement will incorrectly predict 3,3-dimethylbut-1-ene.
Rate Law Identification:
Reveal answer
SN2 or E2. Rate depends on both substrate and base/nucleophile concentration → second-order kinetics → bimolecular mechanisms. Rate = k[substrate][base] fits both SN2 and E2.
Reveal answer
SN1 or E1. Rate depends only on substrate → first-order kinetics → unimolecular mechanisms. The nucleophile/base isn't involved in the rate-determining step (ionization of the substrate). Both SN1 and E1 share this rate law.
Reaction Coordinate Matching:
Reveal answer
SN2 or E2. One transition state with no intermediate = concerted, one-step mechanism. Both SN2 and E2 are concerted and show a single energy barrier. You'd need additional info (does the product have a new bond to a nucleophile, or a new pi bond?) to distinguish between them.
Reveal answer
SN1 or E1. Two transition states with a carbocation intermediate = two-step mechanism. The first step (ionization, forming the carbocation) has the higher barrier because it's rate-determining. Both SN1 and E1 share this energy profile; they diverge only at the second step (nucleophilic attack vs. deprotonation).
Which Reacts Faster (select A or B):
Reveal answer
(A) is faster. SN1 requires carbocation formation. Polar protic solvents (water) stabilize the carbocation through solvation, lowering the activation energy for ionization. Polar aprotic solvents (DMSO) do not stabilize carbocations well. Same substrate, but the solvent in (A) accelerates the rate-determining step.
Reveal answer
(A) is faster. SN2 rate depends on steric accessibility of the electrophilic carbon. 1-bromobutane (primary) has minimal steric hindrance at the reaction center. 2-bromobutane (secondary) has more substituents blocking backside attack. Same nucleophile and solvent, but (A) has less steric hindrance.
Reveal answer
(A) is faster. E2 requires anti-periplanar geometry between the leaving group and the β-hydrogen. When Br is axial, adjacent axial H atoms are anti-periplanar → E2 proceeds readily. When Br is equatorial, no adjacent H is anti-periplanar → E2 is extremely slow. This is the same principle tested in Example 9 with the tert-butylcyclohexane.
SN1 vs SN2 vs E1 vs E2: How to Choose the Right Mechanism is only available for OrgoSolver Pro members
Get unlimited access to our content
Interactive study tools like the Mechanism Solver or Resonance Solver, Study Guides, Lab Reports, and more!